Introduction
The framing that has dominated pancreatic cancer drug development for the better part of two decades positions the disease primarily as a molecular targeting challenge. Identify the right gene, the right protein, the right pathway, engineer a sufficiently potent and selective agent, and the tumor should respond. This framework has produced an enormous volume of preclinical data, a steady stream of clinical trials, and a survival curve that has barely moved.
The problem is not that the targets are wrong, though some of them may be. The problem is that the framework misrepresents the biology it is trying to address. PDAC tumors are not masses of genetically abnormal cells waiting to be eliminated by a sufficiently precise molecular intervention. They are structured ecosystems, actively maintained by a complex community of stromal, immune, and vascular components, each contributing to a biological environment that resists therapeutic pressure with a sophistication that no single target can account for.
The Tumor Ecosystem and Its Active Role in Resistance
The cellular composition of the PDAC tumor microenvironment includes cancer cells, cancer stem cells, and progenitor cells at the tumor core, but extends well beyond them. Cancer-associated fibroblasts produce and remodel the extracellular matrix, regulating the physical properties of the tumor stroma and producing signaling molecules that sustain cancer cell survival. Pancreatic stellate cells, a fibroblast-like population unique to the pancreatic microenvironment, contribute to the desmoplastic reaction that defines the gross pathology of PDAC and drives the dense fibrotic architecture that characterizes the disease.
Tumor-associated macrophages, monocytes recruited from the circulation, and populations of regulatory immune cells create an immunological landscape that actively suppresses anti-tumor immunity. These are not passive components. They are dynamically regulated by signals from the tumor cells and from each other, forming a reciprocally reinforcing system in which the immune environment and the tumor biology sustain one another.
Mesenchymal stem cells and other stromal populations contribute further to this ecosystem by providing metabolic support, secreting growth factors, and participating in the remodeling responses that occur when the tumor is placed under therapeutic stress. The extracellular matrix itself, composed of collagen, fibronectin, hyaluronan, and other structural proteins, is not simply a scaffold. It is a signaling-active environment that communicates mechanical and biochemical information to the cells embedded within it, shaping their behavior in ways that contribute to drug resistance and immune exclusion.
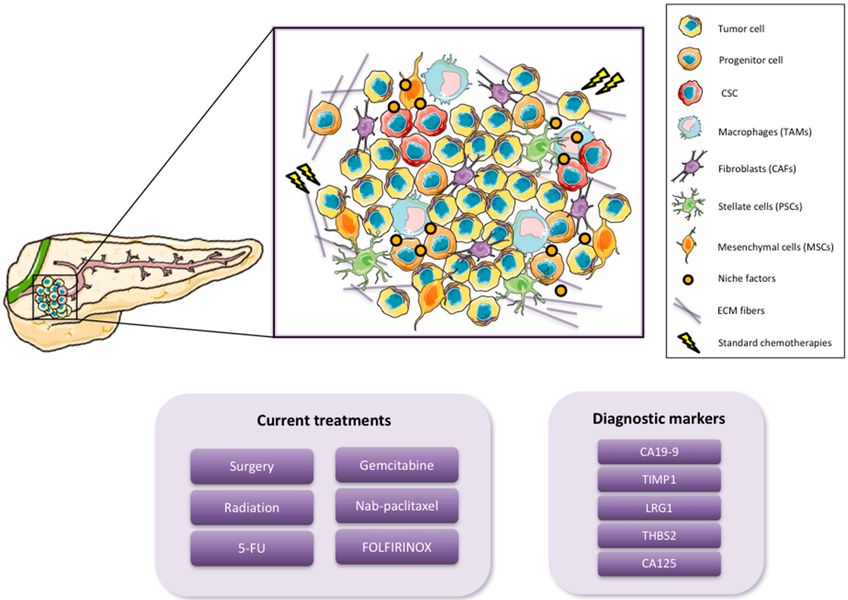
Three Mechanisms by Which the Microenvironment Defeats Therapy
The microenvironment does not resist therapy through a single mechanism. It operates through at least three distinct but interconnected processes that collectively make PDAC one of the most treatment-refractory solid tumors in clinical oncology.
Dense stroma limits drug penetration. The elevated interstitial fluid pressure within PDAC tumors, generated by the dense extracellular matrix and the compressed, disorganized vasculature it creates, opposes the convective transport of systemically administered drugs. Compounds that achieve therapeutic concentrations in the circulation may never reach meaningful intratumoral levels. This is not a pharmacokinetic failure. It is an architectural one, built into the physical structure of the tumor by the CAFs and stellate cells that produce and maintain the stromal matrix.
Immune suppression blocks clearance. The immunosuppressive polarization of tumor-associated macrophages, the exclusion of cytotoxic T cells from the tumor core, and the enrichment of regulatory immune populations collectively ensure that the adaptive immune system cannot eliminate the cancer cells that chemotherapy and targeted therapy leave behind. Therapies that depend on immune-mediated clearance for full efficacy are therefore operating at a structural disadvantage in PDAC regardless of their direct cytotoxic activity.
Metabolic coupling buffers stress. The stromal compartment in PDAC is not metabolically inert. Stellate cells and other stromal populations engage in metabolic exchanges with cancer cells, providing alternative carbon and nitrogen sources that allow the tumor to maintain function under nutrient deprivation and therapeutic stress. This metabolic buffering capacity means that interventions designed to starve the tumor of specific metabolites are met with compensatory supply from the stromal compartment.
Why System-Level Thinking Changes the Therapeutic Question
The practical implication of the ecosystem model is that the relevant unit of therapeutic design is not the cancer cell or any individual target within it. It is the integrated system that the cancer cell depends on to survive. Targeting a single component of this ecosystem, whether a signaling pathway in the cancer cell, a specific stromal marker, or a single immune checkpoint, leaves the compensatory capacity of the remaining ecosystem intact. The system adapts, often more rapidly than the drug can act, and the initial response is followed by resistance.
This does not mean that targeted therapy is futile in PDAC. It means that the design question has to change. Instead of asking which target to inhibit, the relevant question is how to perturb the system in ways that exceed its adaptive capacity across multiple dimensions simultaneously. A compound or combination that imposes stress on the cancer cell while also compromising the microenvironmental support that enables recovery is mechanistically better positioned than one that achieves the same degree of on-target activity without addressing the ecosystem.
Redox perturbation, metabolic disruption, and approaches that alter the biochemical environment on which the entire ecosystem depends represent one avenue toward this kind of system-level engagement. The goal is not a cleaner target. It is a more complete disruption of the conditions under which PDAC survives.
Conclusion
PDAC’s resistance to therapy is not primarily a consequence of inadequate target identification or insufficient drug potency. It is a consequence of targeting single components within a structured, adaptive ecosystem that responds to therapeutic pressure by engaging the compensatory capacity of its other components. Fibroblasts, immune cells, stellate cells, and the extracellular matrix are not bystanders in this disease. They are active participants in the biology of resistance. Therapeutic strategies that do not account for this ecosystem will continue to produce the same pattern of transient response and rapid resistance that has characterized PDAC treatment for decades. The shift from target thinking to system thinking is not a refinement of the existing approach. It is the necessary correction to a framework that has consistently misrepresented the nature of the problem.
