Introduction
The standard model of drug resistance in oncology treats it as something that develops. A drug is administered, an initial response is observed, and over weeks or months the tumor evolves mechanisms to circumvent the therapy. Under this model, resistance is a downstream consequence of treatment, a biological response to selective pressure that appears after the drug has had its opportunity to act.
This model is clinically intuitive. It maps onto what oncologists observe at the bedside: partial responses that fade, stable disease that progresses, and tumors that eventually grow through the most aggressive available regimens. But the model is incomplete as a description of what is actually happening in pancreatic ductal adenocarcinoma, and that incompleteness has consequences for how therapeutic strategies are designed.
In PDAC, resistance is not something that emerges in response to treatment. It is the baseline state of the tumor. Understanding the difference between these two framings, and why it matters for drug development, is the conceptual shift that opens the door to more durable therapeutic approaches.
What Baseline Resistance Actually Means
When oncologists describe a tumor as resistant, they typically mean that it has stopped responding to a specific drug or regimen. The implication is that the tumor was once susceptible and became resistant through an acquired adaptation. In many cancers, this is an accurate description. Cells with pre-existing mutations that confer resistance are selected for during treatment, the sensitive population is eliminated, and the resistant clone expands.
In PDAC, the picture is different. The tumor microenvironment, the metabolic adaptations of the cancer cells, the stromal architecture, and the immunosuppressive landscape are all organized around resistance to therapeutic pressure before any treatment is administered. The fibrotic stroma that limits drug penetration is not produced in response to chemotherapy. The immunosuppressive macrophage polarization that excludes cytotoxic immune cells is not an acquired adaptation to immune checkpoint therapy. These are constitutive features of the disease at diagnosis.
What this means at the molecular level is that PDAC cells in resistant model systems show coordinated activity across multiple adaptive programs simultaneously, not as a consequence of drug exposure, but as a property of their baseline biology. Stress response pathways are upregulated. Metabolic signaling is organized around nutrient scavenging and redox compensation. Transcriptional programs that control survival under stress are active. These are not responses to a drug. They are the baseline operating state of a system that evolved under constitutive selective pressure.
Three Coordinated Shifts That Define Resistant Biology
The most important insight from studying PDAC in genuinely resistant models, rather than in sensitive cell lines that are then exposed to drug until resistance emerges, is that resistance is not a single mechanism. It is a coordinated state involving at least three interconnected adaptive programs that operate simultaneously.
Stress response pathway activity defines one dimension of this state. Resistant PDAC cells show elevated baseline activity in the heat shock response, the unfolded protein response, and other programs that cells normally activate only under acute stress. This constitutive stress response activity provides a buffer against additional insults, including therapeutic stress, that a cell without this baseline activation would not have. When a drug imposes stress on a resistant cell, the stress response machinery is already primed to manage it.
Metabolic signaling organization constitutes a second dimension. Resistant PDAC cells engage in active metabolic reprogramming that is not merely a consequence of the hypoxic, nutrient-poor microenvironment, but a feature of how the cells are intrinsically organized. Autophagy, macropinocytosis, and amino acid scavenging pathways are constitutively active, providing alternative nutrient sources that buffer the cell against metabolic disruption. This metabolic flexibility makes it difficult for any single metabolic intervention to achieve the degree of stress required to push the cell toward death.
Transcriptional control of survival represents the third coordinated shift. Resistant PDAC cells show elevated baseline activity in transcription factors and transcriptional programs that maintain survival under stress, including NF-kB, STAT3, and programs that suppress apoptotic gene expression. These transcriptional states are self-reinforcing and highly stable, meaning that simply blocking one component of the network is unlikely to overcome the survival program as a whole.
Why This Demands a Different Therapeutic Design Principle
The practical implication of baseline resistance as a biological state rather than an acquired response is that drug design must start from the assumption that all three of these adaptive programs are active simultaneously from the first dose. A compound designed to exploit a vulnerability that only appears after resistance develops will never have an opportunity to act on that vulnerability in a PDAC patient, because resistance is already present before treatment begins.
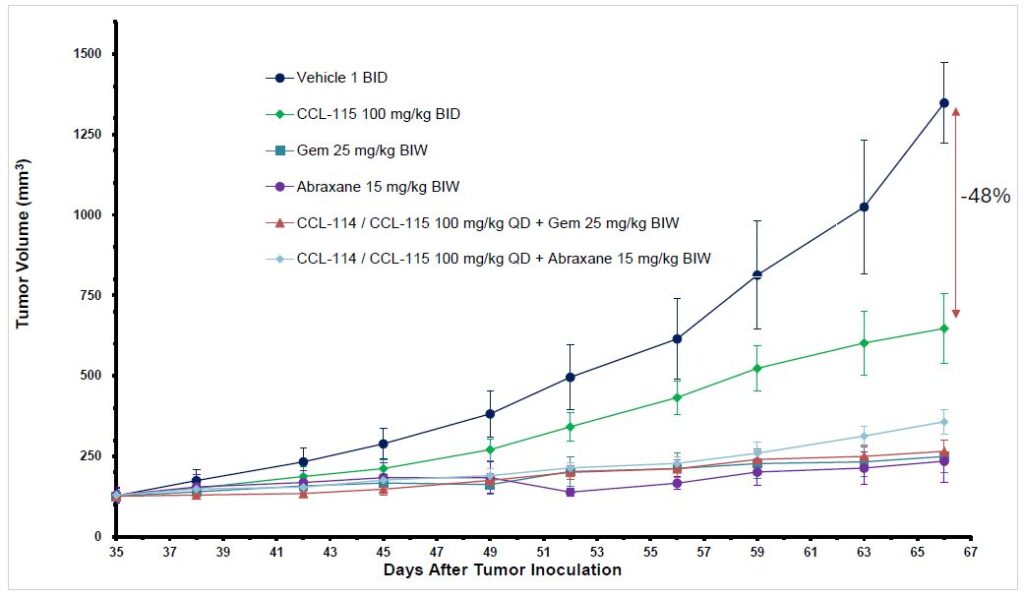
This shifts the design question from how to delay or prevent resistance to how to engage with resistant biology directly and overcome its adaptive capacity. CCL-114 and CCL-115 have demonstrated 48 percent tumor volume reduction versus vehicle control in a resistant PDAC model, in combination with standard of care chemotherapy. The significance of this finding is not simply the magnitude of the reduction. It is that the reduction was achieved in a system where baseline resistance was the operative condition.
Understanding what these compounds do in resistant systems, specifically how they interact with the constitutively active stress response, metabolic, and transcriptional programs that define resistant PDAC biology, is the scientific question that will determine whether the observed efficacy reflects a mechanism capable of sustaining durable clinical benefit or a transient effect that the resistant system will eventually compensate around.
Conclusion
Resistance in PDAC is not a problem that appears after treatment. It is the biological state from which treatment must begin. Coordinated activity across stress response pathways, metabolic signaling, and transcriptional survival control is present at baseline in resistant models, not as an adaptation to drug exposure but as a constitutive feature of how these cells are organized. Drug programs designed around the assumption that resistance is a late-stage event will consistently underperform in PDAC because they are solving the wrong problem. The compounds and strategies that will eventually produce durable benefit in this disease are those designed from the outset to engage with resistant biology as the baseline condition, not the exceptional outcome.
