Introduction
Pancreatic ductal adenocarcinoma is routinely described as one of the most treatment-resistant cancers in clinical oncology. The standard explanation focuses on biology at the cellular level: KRAS mutations, redundant survival signaling, and the adaptive capacity of the cancer cell itself. These are real and important challenges. But they account for only part of the problem.
The other part, and arguably the more underappreciated part, is structural. PDAC tumors are not simply collections of malignant cells waiting to be targeted. They are physically and biologically fortified systems embedded in a dense, actively maintained microenvironment that impedes drug delivery, suppresses immune recognition, and sustains tumor survival independent of the cancer cell’s own adaptive machinery. Understanding why most drugs fail in PDAC requires understanding this architecture, not just the cancer cell it protects.
The Structural Barrier: Stroma and Cancer-Associated Fibroblasts
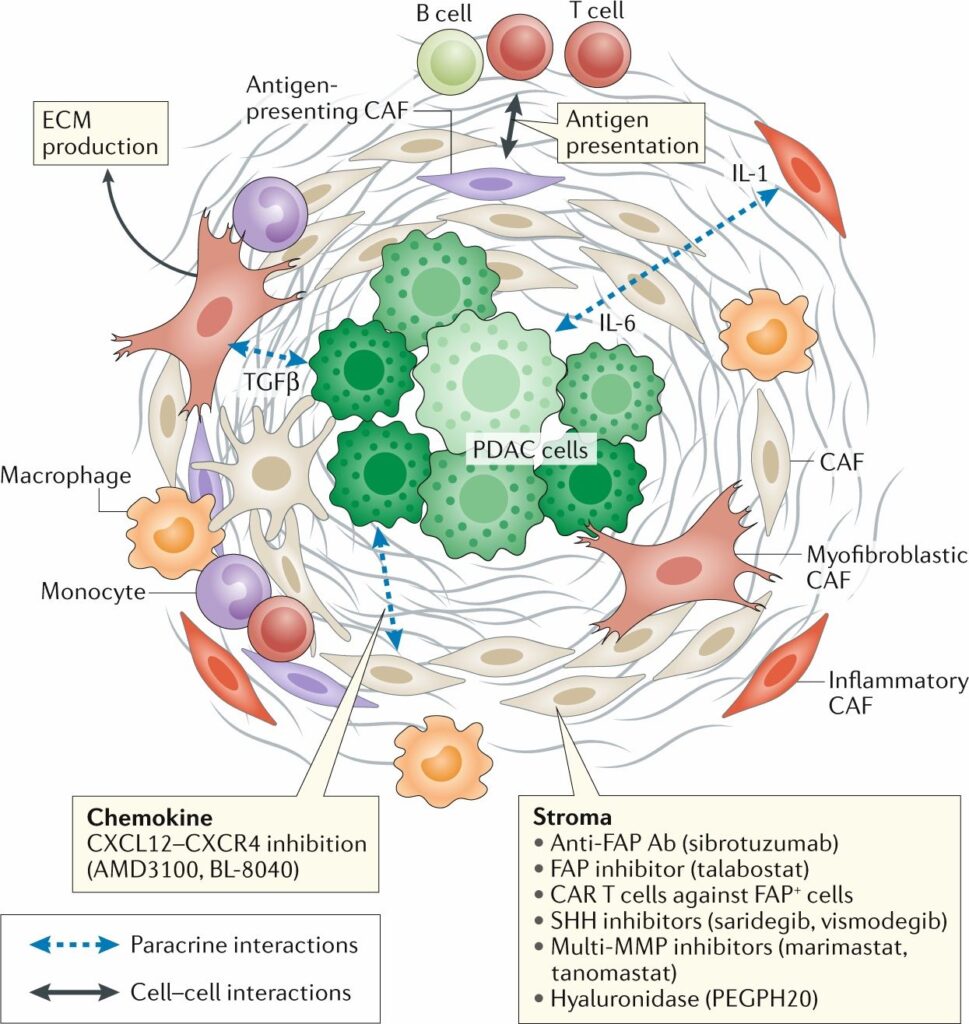
The most immediately consequential feature of the PDAC tumor microenvironment is the fibrotic stroma that surrounds and infiltrates the tumor mass. Unlike the relatively permissive tissue architecture of many other solid tumors, the PDAC stroma is dense, collagen-rich, and actively produced by a heterogeneous population of cancer-associated fibroblasts. These CAFs are not passive bystanders. They are recruited, activated, and maintained by signals from the tumor cells themselves, creating a reciprocal relationship in which the stroma and the cancer reinforce each other’s persistence.
The physical consequences of this architecture for drug delivery are substantial. The interstitial pressure within PDAC tumors is significantly elevated compared to normal tissue, creating a pressure gradient that opposes the convective transport of systemically administered therapeutics into the tumor core. Vasculature within and around PDAC tumors is disorganized, compressed by the surrounding stroma, and functionally compromised in ways that reduce blood flow and drug perfusion. The practical result is that even compounds with strong in vitro activity against PDAC cells may never reach meaningful intratumoral concentrations when administered systemically.
CAFs contribute to this barrier both structurally and functionally. Myofibroblastic CAFs produce the extracellular matrix components that constitute the physical scaffolding of the stroma. Inflammatory CAFs secrete cytokines including IL-6 and IL-1 that sustain the inflammatory state of the microenvironment and promote tumor cell survival. The stroma is not simply an inert barrier. It is a biologically active compartment with its own signaling architecture that converges on the support of tumor persistence.
Immune Exclusion and Immunosuppressive Remodeling
Beyond the physical barrier, the PDAC tumor microenvironment is characterized by profound immune exclusion. Cytotoxic T cells, which represent the primary immune mechanism capable of recognizing and eliminating cancer cells, are present in the periphery of PDAC tumors but largely absent from the tumor core where they would need to operate. This is not accidental. The PDAC microenvironment actively maintains an immunosuppressive state through multiple overlapping mechanisms.
Tumor-associated macrophages in PDAC are predominantly polarized toward an anti-inflammatory phenotype that suppresses cytotoxic immune activity rather than supporting it. Monocytes recruited to the tumor site are similarly reprogrammed by microenvironmental signals, including TGF-beta produced by CAFs, into cells that protect rather than attack the tumor. Regulatory T cells, which normally suppress excessive immune activation, are enriched within PDAC tumors and contribute to the immune-excluded state that makes checkpoint immunotherapy largely ineffective in this disease.
The consequence is a tumor that has effectively edited its immunological context. Immune cells that should recognize and eliminate PDAC cells are present in the broader tissue but are either excluded from the tumor, rendered non-functional within it, or actively reprogrammed to support tumor persistence. Immune checkpoint inhibitors, which have transformed outcomes in several other cancers by releasing the brake on anti-tumor immunity, have shown minimal efficacy in PDAC precisely because this deeper immunosuppressive architecture remains intact regardless of checkpoint status.
Why the System Adapts and What That Demands of Therapeutic Strategy
The most important conceptual implication of the PDAC tumor microenvironment is that it is not static. It is a dynamic, adaptive system that responds to therapeutic pressure in ways that sustain tumor survival. When drug delivery is partially improved, the stroma can remodel in ways that re-establish the barrier. When immune access is partially restored, compensatory immunosuppressive mechanisms can re-exclude cytotoxic cells. The microenvironment behaves as an extension of the tumor’s adaptive capacity, not merely a physical obstacle to be bypassed.
This has direct consequences for therapeutic strategy. Approaches that address single components of the microenvironment sequentially have shown limited success in PDAC because the system compensates through the components that remain unaddressed. Stromal depletion strategies, for example, have in some preclinical settings paradoxically accelerated disease progression by disrupting structural constraints on tumor invasion while leaving immunosuppressive signaling intact.
What the architecture of the PDAC tumor microenvironment demands is intervention at the level of the system rather than any individual component within it. The question is not which stromal target to inhibit or which immune checkpoint to release. It is how to perturb the overall biochemical and physical environment in ways that the tumor’s adaptive machinery cannot compensate for through microenvironmental remodeling.
Conclusion
Pancreatic cancer is not just hard to target at the cellular level. It is physically and biologically shielded by a microenvironment that impedes drug delivery, suppresses immune recognition, and actively maintains tumor survival through signals, structural remodeling, and immunological reprogramming. Most drug strategies in PDAC fail not because they lack potency against cancer cells in isolation, but because they were never designed to engage with the system-level architecture that protects those cells in vivo. Recognizing this distinction is the starting point for a different kind of therapeutic thinking, one that asks not which target to hit but how to disrupt the integrated system that PDAC depends on to survive
