Introduction
Pancreatic cancer remains one of the most lethal and treatment-resistant malignancies in modern oncology. Despite decades of research and significant investment, the five-year survival rate for pancreatic ductal adenocarcinoma (PDAC) continues to hover around 12 percent, a figure that has improved only marginally over the past thirty years. The reasons for this persistent failure are not rooted in a lack of scientific effort. They are rooted in a fundamental misalignment between what drug programs optimize for and what patients actually need to survive.
The dominant approach in early-stage oncology drug development has long prioritized potency, specifically the ability to kill cancer cells efficiently in a controlled laboratory environment. While this metric is scientifically valid, it is increasingly recognized as an insufficient predictor of real-world clinical success. What kills a cell in a dish does not always translate into what eliminates a tumor in a living organism without collateral damage. This gap between in vitro promise and in vivo reality is where the vast majority of oncology programs collapse.
A new class of programs centered on redox biology is beginning to challenge this paradigm. By targeting the oxidative stress vulnerabilities unique to pancreatic cancer cells, and by placing equal emphasis on selectivity, systemic tolerability, and translational robustness, these approaches are attempting to solve the problem that has defined oncology drug development for generations.
The Translation Problem in PDAC Drug Development
To understand why so many PDAC programs fail, it is important to distinguish between activity and efficacy in a clinical sense. Activity refers to the measurable effect of a compound on cancer cells or tumors under experimental conditions. Efficacy refers to whether that effect is meaningful, durable, and achievable in a patient without causing unacceptable harm.
Most compounds that enter preclinical development demonstrate strong activity. They reduce cell viability, induce apoptosis, and suppress proliferation with measurable IC50 values. Laboratories worldwide generate this data reliably and reproducibly. The problem begins when these compounds move into animal models, and becomes critical when they advance to human trials.
PDAC presents a uniquely hostile environment for drug delivery. The tumor microenvironment is characterized by dense stromal tissue, poor vascularization, and a heavily immunosuppressive landscape. These structural features physically impede drug penetration and create conditions under which cancer cells rapidly develop resistance mechanisms. Compounding this is the biological complexity of KRAS-mutant signaling, which drives the majority of PDAC cases and has historically resisted direct therapeutic targeting.
Programs that fail to account for this complexity during their design phase are essentially solving the wrong problem. Optimizing for potency against sensitive cell lines in isolation does not prepare a drug candidate for the metabolic adaptability, stromal shielding, and genetic plasticity that define PDAC in vivo.
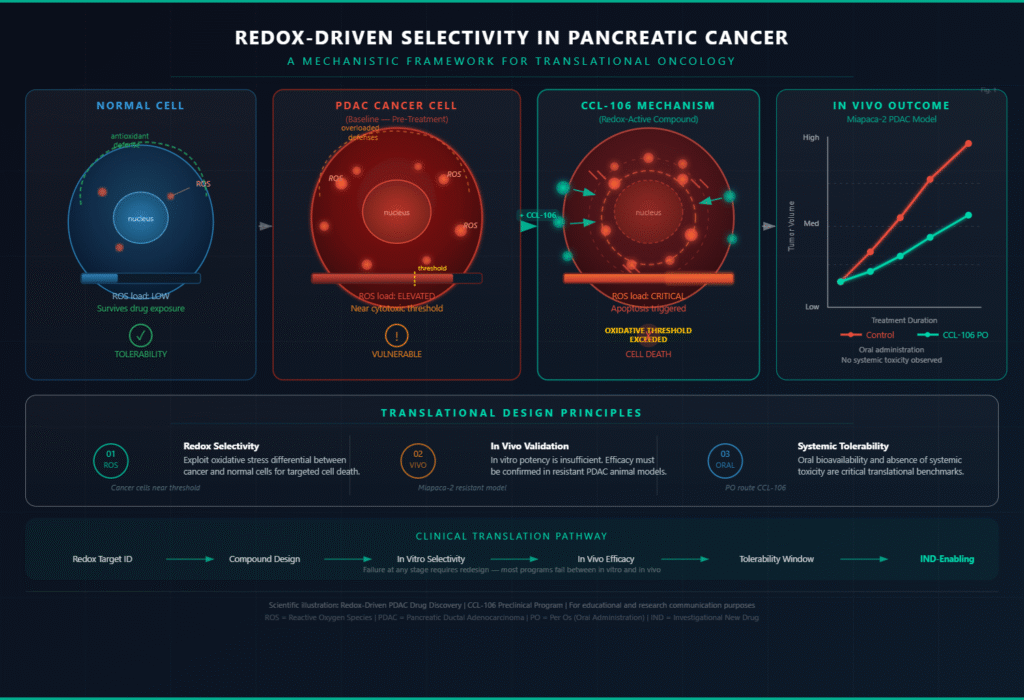
Redox Biology as a Therapeutic Lever in Pancreatic Cancer
Redox biology concerns the balance between oxidative and reductive chemical processes within cells, governed primarily by reactive oxygen species (ROS) and the antioxidant systems that regulate them. What makes this framework particularly relevant to PDAC is a well-documented paradox: pancreatic cancer cells exist in a state of chronically elevated oxidative stress, yet they survive by upregulating antioxidant defenses to prevent ROS levels from reaching a cytotoxic threshold.
This creates a targetable vulnerability. Cancer cells that are already operating near their oxidative stress limit are inherently more susceptible to compounds that further elevate ROS or disrupt the antioxidant systems keeping them viable. Normal cells, which are not under the same degree of oxidative pressure, can tolerate modest increases in ROS without reaching a crisis point. This differential forms the biochemical basis for the selectivity that redox-driven programs seek to exploit.
Redox-active drug candidates designed around this principle do not simply aim to be toxic. They aim to be selectively toxic, pushing cancer cells over an oxidative threshold while remaining within a tolerable range for healthy tissue. This is a mechanistically coherent approach to the selectivity problem that has derailed so many conventional cytotoxic agents. The challenge lies in engineering that selectivity with sufficient precision, and then validating it not just in cell lines but in models that more faithfully reflect human tumor biology.
What In Vivo Data Reveals That In Vitro Cannot
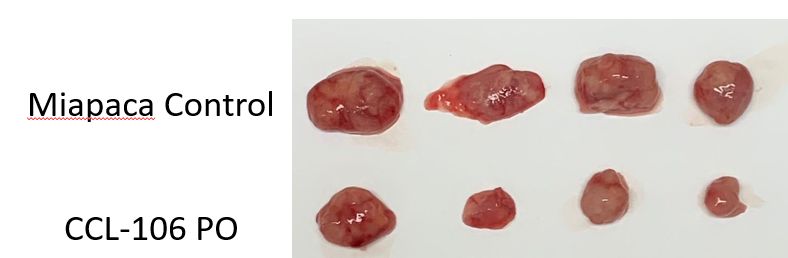
The image at the center of this discussion, showing excised tumor specimens from a Miapaca-2 pancreatic cancer model comparing untreated control animals against those receiving an orally administered compound designated CCL-106, illustrates something that no cell viability assay can: physical, measurable tumor reduction in a living system.
Miapaca-2 is a well-characterized PDAC cell line known for its aggressive growth profile and resistance to standard-of-care agents. Its use as a model system carries scientific weight precisely because it is not a sensitive or cooperative target. Tumor reduction in this context, visible without magnification across multiple animals, is a meaningful preclinical signal.
Oral bioavailability adds a further dimension of clinical relevance. Intravenous delivery of oncology compounds, while common in research settings, introduces variables related to formulation, administration tolerance, and patient burden that frequently complicate clinical translation. An orally administered agent that retains activity in a resistant PDAC model addresses one of the core practical barriers to real-world use.
The absence of obvious systemic toxicity at active doses, while requiring further characterization through formal safety studies, suggests a tolerability window that many cytotoxic oncology programs never achieve. This does not constitute safety validation, but it represents exactly the kind of early signal that distinguishes candidates worth advancing from those that show potency alone.
The Design Philosophy That Changes Outcomes
What separates translatable oncology science from laboratory curiosity is not the sophistication of the chemistry alone. It is the design philosophy that governs which questions are asked at each stage of development. Programs that begin by asking what kills these cells most efficiently are structurally predisposed to generating potent but non-selective compounds that accumulate toxicity before they accumulate benefit.
Programs that begin instead by asking where, how, and at what cost does this compound exert its effect are positioned to generate candidates that can survive the rigors of translation. Selectivity, tolerability, and mechanistic coherence are not features added later in development. They are properties that must be engineered in from the outset.
The redox-driven approach to PDAC represents this kind of principled design. It acknowledges the biological realities of the disease, including its resistance mechanisms, its microenvironmental barriers, and its metabolic dependencies, and builds a strategy that engages with those realities rather than ignoring them. It measures success not by how efficiently a compound clears a cell culture well, but by whether it can be administered, tolerated, and effective in the context of a living organism with an intact physiology.
Conclusion
The history of pancreatic cancer drug development is, in many respects, a history of failed translations. Compounds that looked promising in vitro have repeatedly proven inadequate in vivo, and candidates that showed early clinical signals have collapsed under the weight of systemic toxicity or acquired resistance. This is not a failure of chemistry. It is a failure of framework.
Redox-targeted programs that prioritize selectivity and in vivo tolerability alongside activity represent a structurally different approach to this problem. The preclinical data emerging from programs like the one described here, particularly visible tumor reduction in resistant PDAC models with oral delivery and an early favorable tolerability profile, reflects what the field needs more of: science designed for the patient, not just for the assay.
The challenge of translating oncology science from bench to bedside is not unsolvable. It simply requires asking the right questions at the right time, and refusing to define success by metrics that do not ultimately predict survival.

