Fractional purification in chemical manufacturing is frequently perceived as a journey that culminates with the precision of chromatography. However, for the industrial chemist, the true terminal step of isolation is not the separation of compounds, but the physical filtration of the resulting mixture. While laboratory scale operations often treat filtration as a minor procedural footnote, the transition to production scale reveals it as the primary determinant of throughput and economic viability. In this environment, the objective shifts from merely collecting a chemical entity to managing a complex mechanical interface where flow rate dictates the success of the entire synthesis.
Mechanical Realities and the Physics of Industrial Filtration
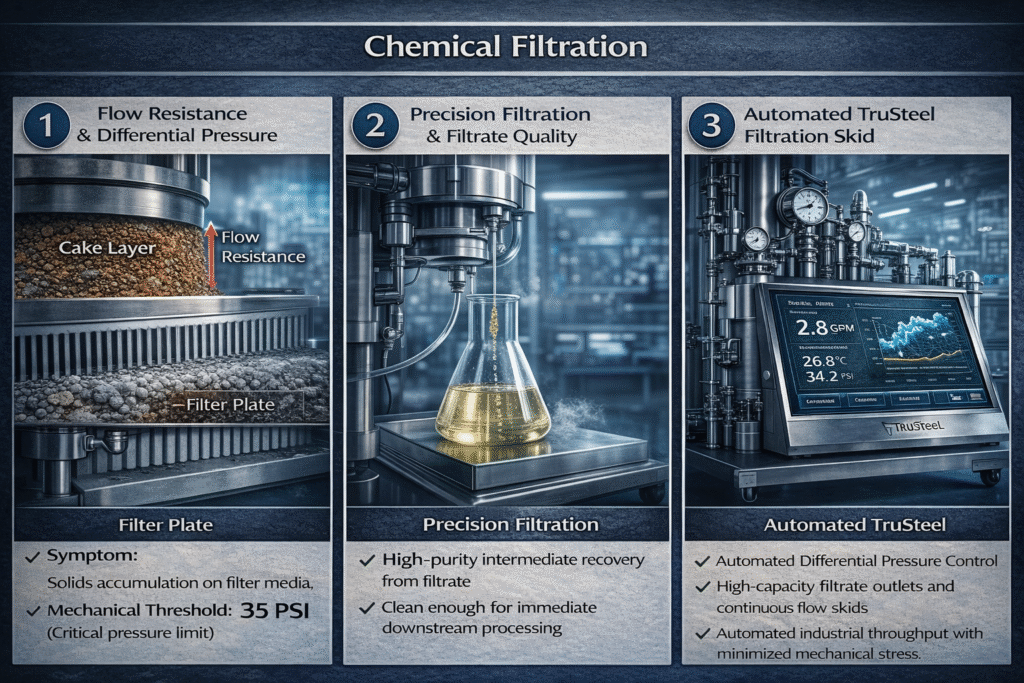
At the industrial level, the chemical yield of a reaction often takes a back seat to the physical characteristics of the solid phase. The “crystal habit”—the characteristic external shape of an individual crystal—becomes more significant than the theoretical purity of the sample. If the solids form “fines” or irregular shapes that create a highly resistive cake, the process slows to an unsustainable pace. This is where scale provides its most difficult lessons: “high purity” as a chemical metric is essentially meaningless if the physical flow rate of the filtrate collapses under the weight of an unmanageable solids load.
The engineering challenge lies in the accumulation of solids, which do not simply “filter out” but instead create a dynamic barrier that increases resistance over time. In production environments, filtration becomes a critical throughput decision. If the differential pressure across the filter media exceeds mechanical thresholds, the process fails quietly through blinded plates and increasingly slow cycles. This attrition is a primary mode of failure in fractional purification in chemical manufacturing, where the inability to isolate an intermediate efficiently can stall an entire downstream workflow.
Filtrate Recovery and Downstream Integrity
In many modern extraction processes, particularly those targeting botanical or complex organic scaffolds, the true value resides in the filtrate rather than the filter cake. The solids, often composed of spent plant matter or synthetic debris, are merely a barrier to be cleared so that the liquid phase can move toward concentration and final refinement. Recovering this filtrate with absolute clarity is essential for protecting sensitive downstream equipment, such as continuous solvent recovery systems or falling film evaporators.
Any particulate matter or “fines” that escape the primary filtration stage can lead to cumulative mechanical failures in pumps and seals. This is why the filtration strategy must prioritize clean filtrate recovery to ensure that subsequent steps, like solvent removal, can operate at peak efficiency without the risk of fouling or thermal degradation. By treating the filtrate as the primary product, manufacturers can optimize their fractional purification in chemical manufacturing to produce higher-fidelity compounds with reduced waste.
Human and Hardware Limits in Scale Up Operations
When a process transitions from 1 liter to 50 liters or higher, the human element often becomes the “limiting reagent.” The complexity of managing high-pressure filtration skids and automated recovery units requires specialized expertise that goes beyond benchtop chemistry. Hardware like the TruSteel AV30-02 integrates automated monitoring to assist operators in making real-time decisions regarding pressure and flow rates.
Strategic infrastructure, such as automated continuous solvent recovery systems, allows for the data-driven management of these processes. Instead of relying on manual inspections, localized control panels provide immediate feedback on system health, identifying blinded plates or seal creep before they cause a total production halt. This level of control is necessary for maintaining a competitive edge in high-throughput chemical industries where every second of downtime directly impacts the bottom line.
Conclusion
The evolution of a chemical project from the laboratory to the factory floor is marked by the recognition that filtration is the final arbiter of success. Mastering fractional purification in chemical manufacturing requires a balanced focus on chemical kinetics and the mechanical physics of filtration. By prioritizing the recovery of high-quality filtrate and utilizing robust, automated infrastructure to manage the physical challenges of scale, industry leaders can ensure their chemical innovations reach the market without being sidelined by hardware limitations. The future of industrial refinement lies in the seamless integration of chemistry with resilient engineering.

