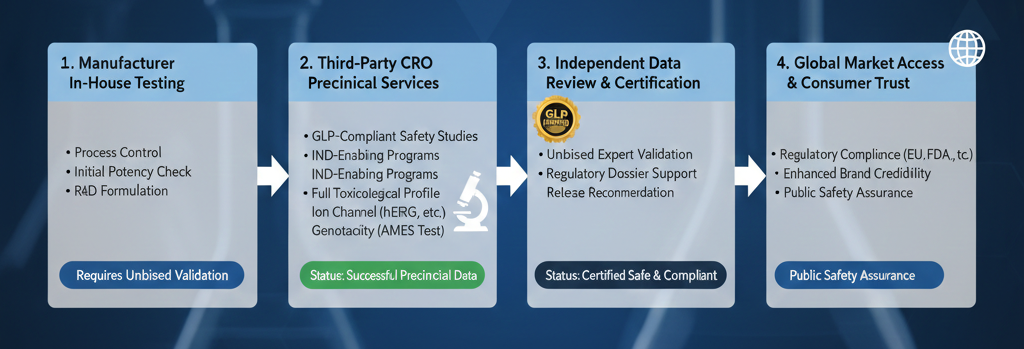
The rapid expansion of the hemp-derived cannabinoid market has outpaced the development of standardized regulatory frameworks, leading to a critical gap in safety verification. While many manufacturers emphasize the quality of their products through internal laboratory results, the industry is reaching a maturity point where self-assessment is no longer sufficient to guarantee consumer safety or regulatory approval. Transitioning toward a model centered on third-party Contract Research Organizations (CROs) is essential for establishing the level of scientific integrity required for pharmaceutical-grade development. This shift ensures that safety claims are backed by unbiased, expert validation, which is the cornerstone of public trust and global market access.
Mitigating Conflict of Interest through Impartial Validation
Internal testing, while a valuable tool for real-time process control and formulation development, inherently carries the risk of institutional bias. When a company is responsible for both the production and the safety certification of a compound, the potential for a conflict of interest can undermine the perceived reliability of the data. Third-party testing serves as a vital “gold standard,” providing an objective distance between the manufacturer and the analytical results.
Contract Research Organizations like Charles River Laboratories or HD Biosciences are designed to operate as impartial entities. Their business model is built upon the accuracy and reproducibility of their data rather than the commercial success of a specific product. By utilizing external preclinical services, developers obtain a rigorous assessment that is free from commercial pressure. This impartiality is not just a preference; it is a fundamental requirement for any compound intended for clinical trials or highly regulated consumer markets, as it provides a defensible data set that can withstand intense scrutiny from both regulators and the scientific community.
IND-Enabling Programs and Preclinical Expertise
The journey from a novel cannabinoid molecule to a market-ready therapeutic requires a complex series of Investigational New Drug (IND) enabling studies. Professional CROs offer comprehensive programs that go far beyond simple potency testing. These services encompass both in vitro and in vivo evaluations designed to map the complete toxicological and pharmacological profile of a substance. This includes assessing systemic toxicity, genotoxicity, and safety pharmacology, particularly regarding cardiovascular and respiratory health.
Expertise in these areas allows for the identification of potential safety signals early in the development cycle. For instance, evaluating how a new cannabinoid interacts with specific ion channels or metabolic enzymes requires specialized equipment and veteran toxicologists that many in-house labs do not possess. By leveraging the infrastructure of established CROs, cannabinoid researchers can access high-fidelity data that supports robust regulatory submissions across various sectors, including pharmaceuticals, medical devices, and even agrochemicals.
Aligning with Global Regulatory Standards and Compliance
As the cannabinoid industry seeks to enter global markets, the complexity of compliance increases exponentially. Different jurisdictions have vastly different requirements for safety data, and a “one size fits all” approach to in-house testing rarely suffices. Professional CRO services are adept at navigating these diverse regulatory landscapes, ensuring that testing protocols align with international Good Laboratory Practice (GLP) standards.
Global standards demand thoroughness and transparency. Aligning with an external testing standard is imperative for any organization looking to export products or obtain international medical certifications. The rigorous documentation provided by a third-party CRO ensures that a product’s safety profile is recognized by international bodies, facilitating smoother entry into European, Asian, and South American markets. This level of compliance is the key to transitioning cannabinoids from a niche agricultural product to a globally recognized therapeutic category.
Conclusion
The future of cannabinoid safety lies in the move toward transparency and impartial validation. While in-house testing remains a necessary component of the manufacturing process, it must be viewed as a preliminary step rather than the final word on safety. Advocating for third-party CRO testing is an advocacy for consumer well-being and scientific excellence. By embracing the expertise of established preclinical leaders, the cannabinoid industry can uphold the highest safety standards and foster a marketplace built on verified, unbiased data. Establishing this dialogue today is the only way to ensure the long-term viability and credibility of cannabinoid-based innovations.

