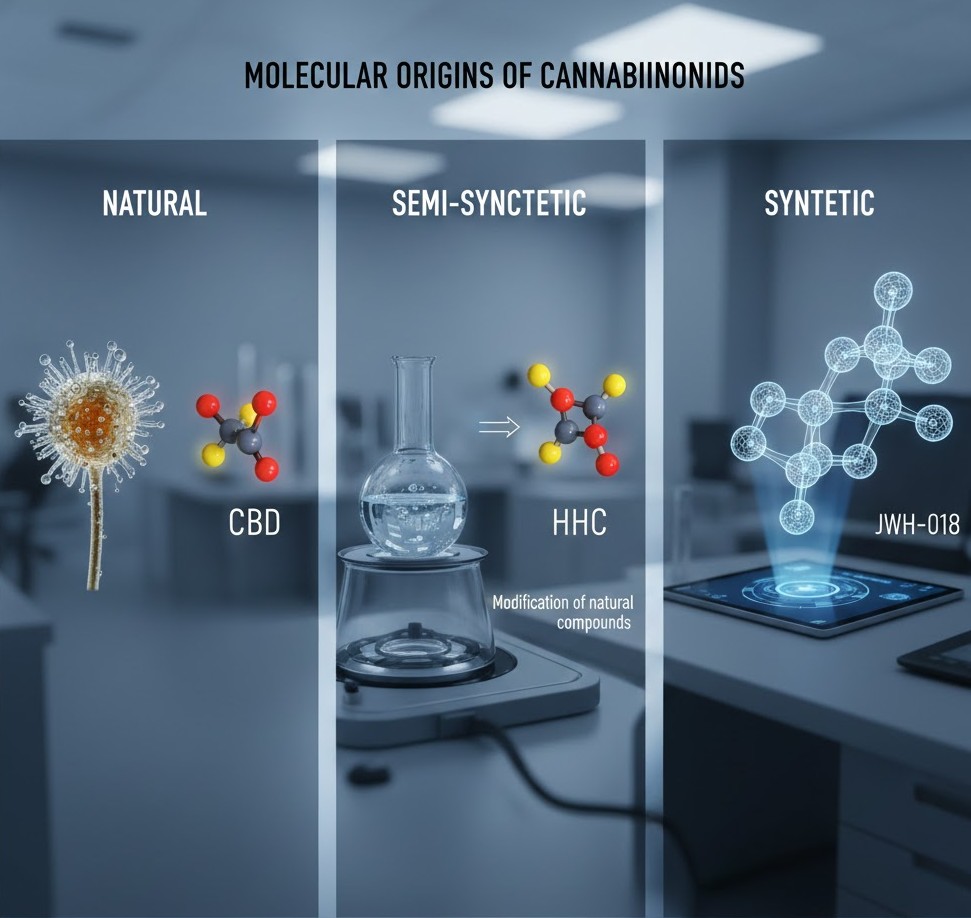
The pharmacological landscape of cannabinoids has expanded far beyond the simple identification of phytocannabinoids found in the resin glands of the cannabis plant. As medicinal chemistry and industrial biotechnology advance, the classification of these compounds based on their origin has become essential for researchers, regulators, and healthcare providers. Understanding whether a molecule is natural, semi-synthetic, or fully synthetic is not merely a matter of nomenclature; it defines the compound’s legal status, metabolic pathway, and predictable safety profile. This article examines the structural and origins-based differences that categorize the three primary tiers of cannabinoid compounds.
Phytocannabinoids and the Complexity of Natural Extraction
Natural cannabinoids, scientifically referred to as phytocannabinoids, are organic compounds produced within the glandular trichomes of the cannabis plant. These molecules, such as Delta 9 tetrahydrocannabinol (THC) and cannabidiol (CBD), are synthesized via complex enzymatic pathways starting from precursor fatty acids and terpenes. In their natural state, these compounds exist as part of a biological matrix that includes hundreds of other secondary metabolites, including flavonoids and aromatic terpenes.
From a research perspective, natural cannabinoids are often valued for the “entourage effect,” a theory suggesting that the therapeutic efficacy of a plant extract is greater than the sum of its individual parts. However, for pharmaceutical applications, the challenge lies in the inherent variability of plant material. Factors such as soil composition, UV exposure, and harvest timing can alter the ratio of these compounds. Therefore, high-performance liquid chromatography (HPLC) is strictly required to ensure that natural extracts meet the rigorous standardization necessary for clinical use.
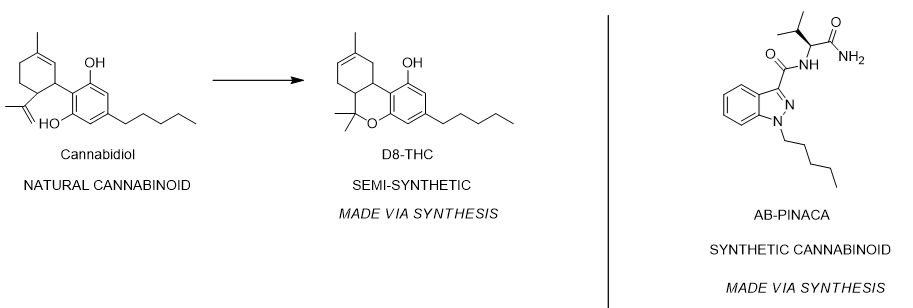
Structural Modification and the Rise of Semi-Synthetic Derivatives
Semi-synthetic cannabinoids bridge the gap between nature and the laboratory. These compounds begin as a natural precursor, typically CBD or THC extracted from hemp or cannabis, which then undergoes specific chemical reactions to alter its molecular structure. The goal of semi-synthesis is often to enhance the potency, stability, or bioavailability of the original molecule. By adding or rearranging functional groups, chemists can create derivatives that may interact more effectively with the CB1 or CB2 receptors.
A prominent example of semi-synthesis is the conversion of CBD into various isomers or the creation of hydrogenated versions of THC. These modifications are meticulously controlled in laboratory settings to ensure that the resulting semi-synthetic molecule retains the core scaffold of the plant derived precursor while gaining improved pharmacological properties. This area of chemistry is particularly important for developing targeted treatments where the natural form of a cannabinoid may be metabolized too quickly or lack the necessary binding affinity for a specific therapeutic outcome.
Laboratory Precision in Fully Synthetic Cannabinoid Design
Fully synthetic cannabinoids are molecules engineered in a laboratory without any natural plant starting material. These compounds are designed to mimic the effects of phytocannabinoids by targeting the same endocannabinoid system receptors, but their chemical structures are often entirely distinct from those found in the cannabis plant. Originally developed for research purposes to map receptor sites, synthetic cannabinoids allow scientists to study the specific mechanisms of action within the human body.
Compounds such as JWH 018 or various aminoalkylindoles represent the synthetic category. Unlike their natural counterparts, synthetic cannabinoids can be designed to be full agonists at cannabinoid receptors, meaning they produce a maximum biological response. This high level of potency, while useful for specific research models, also introduces significant safety concerns if used outside of controlled clinical environments. In the industrial and pharmaceutical sectors, full synthesis is valued for its ability to produce highly pure, consistent compounds at scale, completely independent of agricultural cycles.
Conclusion
The distinction between natural, semi-synthetic, and synthetic cannabinoids is a fundamental pillar of modern cannabinoid science. While natural compounds offer a complex biological profile, semi-synthetic and synthetic versions provide the precision and consistency required for advanced drug development and structural research. As the industry continues to professionalize, the ability to categorize these molecules based on their molecular origin will remain vital for ensuring product safety, efficacy, and regulatory transparency. Navigating this evolving landscape requires a deep appreciation for the chemistry that defines each compound’s journey from its origin to its biological target.