Decoding Chemical Bonds and Reaction Mechanisms Through Infrared Radiation
In the pursuit of pharmaceutical-grade consistency and deep molecular understanding, the cannabis industry has adopted Fourier Transform Infrared Spectroscopy (FT-IR) as a cornerstone for material characterization. FT-IR is a sophisticated analytical technique that identifies organic and inorganic compounds by measuring how their molecular bonds vibrate when exposed to infrared radiation. Unlike other methods that may focus on electronic transitions, FT-IR provides a direct “mechanical” profile of the molecule. This allows scientists to distinguish between various functional groups and even subtle isotopic changes. As research moves toward personalized cannabinoid therapeutics, the ability to utilize FT-IR for both structural elucidation and mechanistic studies via deuteration has become an invaluable asset for the modern laboratory.
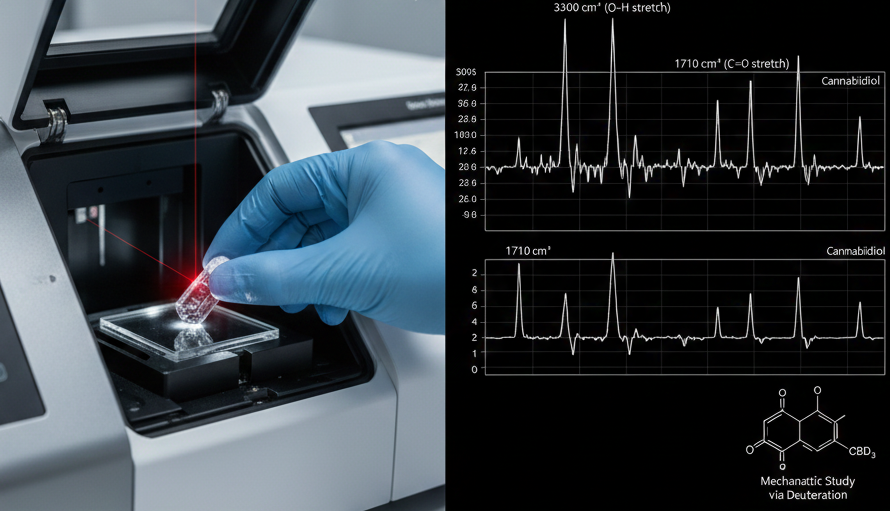
The Physics of Molecular Vibration and Spectral Interpretation
At its core, FT-IR spectroscopy relies on the fact that chemical bonds are not rigid sticks but rather behave like springs with specific vibrational frequencies. When a cannabinoid sample is irradiated with infrared light, the molecules absorb energy at frequencies that match their natural vibrational modes. These vibrations generally manifest as stretching, bending, or twisting of the atomic bonds. In a standard mid-infrared scan, typically ranging from 4000 to 400 reciprocal centimeters (cm^-1), the instrument records which frequencies are absorbed and which are transmitted.
The resulting infrared spectrum is essentially a molecular fingerprint. High-frequency regions often correspond to the stretching of light atoms, such as the O-H and C-H bonds found in CBD and THC. The middle regions, often referred to as the “fingerprint region,” contain complex patterns unique to the specific skeletal structure of the cannabinoid molecule. By identifying these characteristic peaks, chemists can confirm the presence of key functional groups like carbonyls (C=O) or aromatic rings, providing immediate qualitative proof of the sample’s identity and structural integrity.
Advanced Applications in Quantitative Analysis and Formulation
While FT-IR is frequently celebrated for its qualitative strengths, its capacity for quantitative analysis is equally vital in industrial settings. By applying the Beer-Lambert Law, researchers can correlate the intensity or area of specific absorption bands to the concentration of a target cannabinoid within a complex mixture. This is particularly useful in the analysis of cannabis extracts, distillates, or final pharmaceutical formulations where rapid, non-destructive testing is required to ensure potency and uniformity across production batches.
Sample preparation plays a critical role in achieving these precise measurements. Whether a sample is prepared as a thin liquid film or pressed into a potassium bromide (KBr) pellet, ensuring a uniform path length is essential for reproducible results. In modern high-throughput environments, Attenuated Total Reflection (ATR) accessories allow for the direct analysis of solids and liquids with minimal preparation, streamlining the workflow for quality control teams who must verify that products meet rigorous regulatory standards for chemical composition.
Deuteration and the Kinetic Isotope Effect in Mechanistic Studies
A more advanced frontier in cannabinoid chemistry involves the use of deuteration—the process of replacing standard hydrogen atoms with deuterium, a heavier isotope. When deuterium is incorporated into a molecule, the vibrational frequency of the affected bond shifts to a lower frequency due to the increased mass. FT-IR is the primary tool used to monitor these shifts, allowing researchers to confirm successful isotopic labeling. This is fundamentally important for studying the Kinetic Isotope Effect (KIE), which provides deep insights into the rate-determining steps of chemical or biological reactions.
By analyzing how the reaction rate changes when a C-H bond is replaced by a C-D bond, scientists can elucidate the complex pathways of cannabinoid metabolism and biosynthesis. These studies offer a window into how the human body processes compounds in vivo, significantly impacting our understanding of pharmacokinetics and biological activity. Utilizing FT-IR to track these changes allows for a more granular understanding of how semi-synthetic cannabinoids are transformed, ultimately guiding the design of more effective and safer therapeutic agents.
Conclusion
Fourier Transform Infrared Spectroscopy stands as a versatile and indispensable technology in the cannabinoid space. From the basic identification of functional groups to the sophisticated study of reaction kinetics through deuteration, FT-IR provides a level of molecular detail that is essential for innovation. As the industry continues to professionalize, the integration of infrared spectroscopy into standard research and development protocols ensures that the next generation of cannabinoid products is built upon a foundation of structural clarity and mechanistic precision.

