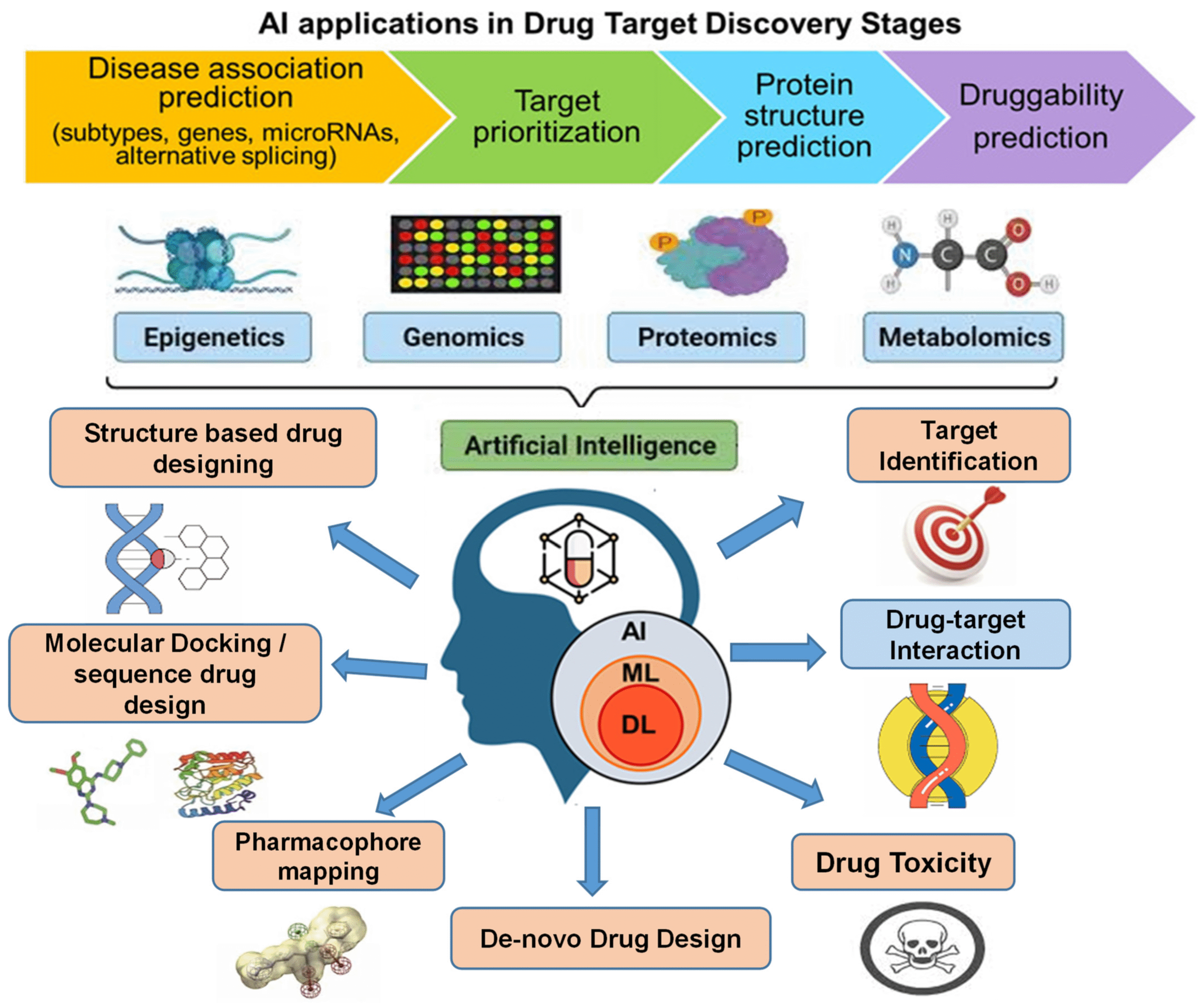
Introduction
Structure activity relationship analysis has long been a cornerstone of medicinal chemistry, guiding scientists in understanding how molecular modifications influence biological activity. Traditionally, this process demands significant manual effort, expert intuition, and extended timelines to design, evaluate, and refine analog libraries. Recent advances in computational chemistry and artificial intelligence are fundamentally reshaping this workflow. Modern platforms now enable rapid generation and evaluation of thousands of molecular variants, allowing researchers to focus more on scientific interpretation and decision making rather than repetitive design tasks. These capabilities are redefining productivity standards in early stage drug discovery.
The Evolution of SAR in Computational Chemistry
SAR analysis originated as a largely empirical exercise, where chemists synthesized small numbers of analogs and evaluated trends over multiple experimental cycles. With the integration of computational modeling, SAR evolved into a more predictive discipline. Algorithms can now assess physicochemical properties, binding interactions, and structural compatibility with biological targets before synthesis begins. This evolution has dramatically reduced development timelines while improving the quality of lead optimization decisions. The shift toward automated SAR generation reflects a broader transformation in how computational tools support hypothesis driven research.
AI Driven Analog Generation and Binding Prediction
The introduction of AI powered plugins has taken SAR analysis to an entirely new level. Using tools such as StarDrop and its advanced design modules, researchers can automatically generate extensive analog libraries from a single lead scaffold. Instead of manually designing dozens of compounds over several days, AI systems can propose hundreds or even thousands of chemically reasonable variants in a matter of hours. These systems also provide predicted binding metrics, enabling rapid prioritization of candidates that are more likely to succeed experimentally. This approach significantly enhances exploration of chemical space while maintaining scientific rigor.
Learning Curves and Research Impact
While AI driven tools lower technical barriers, they also introduce a learning curve for researchers transitioning from traditional workflows. Gaining confidence in interpreting computational outputs and integrating them with experimental insight is a critical step. However, this learning investment yields substantial returns. Rapid analog generation and binding evaluation allow teams to identify structure activity trends earlier, reduce costly synthesis cycles, and make data informed decisions with greater confidence. Over time, these tools empower researchers to become more efficient and more strategic in their scientific reasoning.
Conclusion
AI enabled SAR platforms represent a transformative shift in medicinal chemistry and computational drug design. By automating analog generation and binding prediction, researchers can dramatically accelerate lead optimization while preserving scientific depth. As familiarity with these tools grows, they are likely to become indispensable components of modern research environments, enabling faster discovery, deeper insight, and more informed innovation across the pharmaceutical and chemical sciences.