From Photons to Pharmacopoeia: Leveraging Electromagnetic Interaction for Quality Assurance
As the cannabinoid industry pivots toward pharmaceutical rigor, the demand for accessible yet highly reliable analytical methods has never been higher. Ultraviolet-Visible (UV-Vis) spectroscopy represents one of the most fundamental yet powerful tools in the chemist’s arsenal for the characterization of organic compounds. By measuring how molecules interact with specific wavelengths of light, UV-Vis spectroscopy provides a direct window into the electronic configuration and concentration of a sample. This technique is indispensable for rapid testing, purity verification, and method development, acting as a critical gateway for quality control in the production of high-purity isolates and standardized formulations.
Fundamental Principles of Electronic Transition and Light Absorption
The core of UV-Vis spectroscopy lies in the principle of electronic excitation. When a cannabinoid molecule is exposed to ultraviolet (200 to 400 nm) or visible (400 to 800 nm) light, the valence electrons within its chemical bonds absorb specific amounts of energy. This energy causes the electrons to transition from a low-energy ground state to a higher-energy excited state. Specifically, cannabinoids often contain “chromophores”—functional groups such as conjugated double bonds and phenolic rings—that are highly efficient at absorbing these photons.
The result of this interaction is an absorption spectrum, which serves as a molecular signature. According to the Beer-Lambert Law, the amount of light absorbed is directly proportional to the concentration of the absorbing species and the distance the light travels through the sample. For a laboratory researcher, this mathematical relationship allows for the transition from a simple light measurement to a precise determination of molecular density, providing a rapid assessment of the sample’s chemical profile without the need for extensive destructive testing.
Instrumentation and Quantitative Methodology in the Laboratory
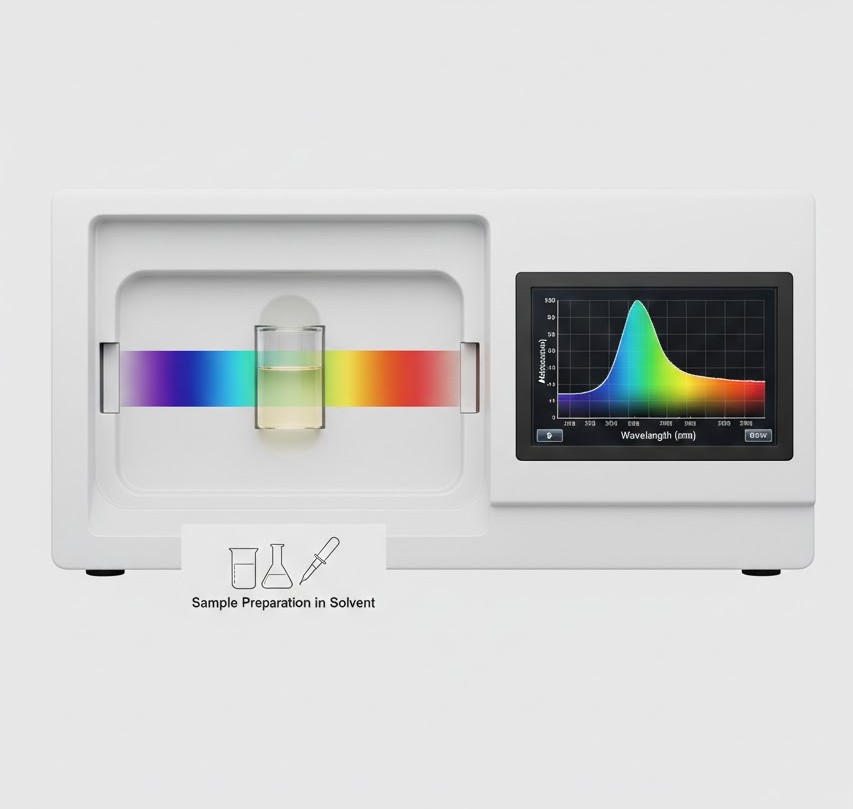
To execute a UV-Vis measurement, the sample is typically dissolved in a spectroscopic-grade solvent, such as ethanol or methanol, and placed into a cuvette made of quartz (which does not absorb UV light). The spectrophotometer directs a beam of light through the sample, while a detector measures the intensity of the light that emerges on the other side. By sweeping across a range of wavelengths, the instrument identifies the Lambda Max—the specific wavelength where the molecule exhibits its highest level of absorption.
In the cannabinoid space, quantitative analysis is a primary application. By constructing a calibration curve using known reference standards, technicians can determine the concentration of a target cannabinoid in an unknown solution with high repeatability. This is particularly useful for monitoring the consistency of distillates during the manufacturing process. While it may not provide the same level of resolution as High-Performance Liquid Chromatography (HPLC) for complex mixtures, its speed and ease of use make it an ideal choice for high-throughput environments where rapid feedback is essential for process optimization.
Purity Assessment and Method Development Protocols
Beyond simple quantification, UV-Vis spectroscopy serves as a vital tool for purity assessment and the identification of contaminants. Pure cannabinoids exhibit characteristic absorption peaks; the presence of “shoulder” peaks or unexpected absorption at different wavelengths can signal the presence of impurities, residual solvents, or degradation products. For example, the oxidation of THC into CBN can be monitored through shifts in the UV spectrum, allowing researchers to track the stability of a product over time under various storage conditions.
Furthermore, UV-Vis is a foundational component of analytical method development. Before more complex techniques like LC-MS are employed, UV-Vis is often used to determine the optimal detection wavelengths for a new compound. Researchers utilize these spectroscopic insights to select appropriate mobile phases and solvent systems, ensuring that the final analytical method is both robust and sensitive. As regulatory requirements for the cannabis industry become more stringent, the role of UV-Vis in establishing validated, reproducible testing protocols continues to expand.
Conclusion
UV-Vis spectroscopy remains a cornerstone of analytical chemistry in the cannabinoid industry due to its versatility, cost-effectiveness, and reliability. By harnessing the interaction between light and electronic structures, scientists can ensure that every batch of product meets the necessary standards for concentration and purity. As we continue to refine our understanding of cannabinoid molecular behavior, the integration of UV-Vis spectroscopy with other advanced analytical platforms will remain essential for driving innovation and ensuring consumer safety in an increasingly complex market.