Fractional purification in chemical manufacturing is often mischaracterized as a purely mechanical or synthetic challenge that occurs within the reactor. However, the true failure point of most industrial processes is not the chemistry itself, but the lack of high-resolution analytical data. When a project transitions from the laboratory bench to multi-kilo campaigns, the margin for error narrows significantly. At this magnitude, qualitative assessments such as “pretty pure” are no longer sufficient specifications for professional production. To achieve consistent results, facilities must invest in advanced instrumentation like Quadrupole Time-of-Flight (Q-TOF) mass spectrometry to identify the minute variables that govern molecular behavior at scale.
High Resolution Analytics and the Science of Trace Impurities
In the context of fractional purification in chemical manufacturing, the ability to detect and quantify trace impurities is the difference between a successful batch and a costly operational failure. While standard chromatography may show a single peak, high-resolution analytics reveal the hidden complexities within that peak, such as structural isomers or unknown byproducts. These trace elements often possess different binding affinities or metabolic profiles, which can compromise the safety and efficacy of the final chemical entity.
The integration of Q-TOF technology allows researchers to establish a precise purity fingerprint for every fraction. By identifying specific degradation pathways and unknown contaminants, engineers can adjust their process parameters in real-time. This level of visibility is essential for ensuring that high-potency analogs remain stable throughout the entire production lifecycle, from initial isolation to final formulation. Without this data, process chemistry remains a form of guesswork that becomes increasingly expensive as volumes grow.
Controlling Molecular Variables for Reliable Scaling
Successful scaling requires the absolute control of every molecular variable, and that control is entirely dependent on data clarity. If an impurity cannot be seen, it cannot be managed; and if it cannot be managed, the process cannot be safely scaled. In fractional purification in chemical manufacturing, unknowns matter because they can act as catalysts for unwanted side reactions or lead to cumulative mechanical failures in sensitive hardware.
Advanced analytical verification ensures that the “crystal habit” and structural integrity of the compound are preserved during high-throughput cycles. By utilizing automated hardware like the TruSteel AV30-02, facilities can link their analytical findings with precise mechanical control. This integration allows for the optimization of vacuum levels and thermal gradients based on the specific stability requirements of the molecule, effectively bridging the gap between theoretical research and industrial execution.
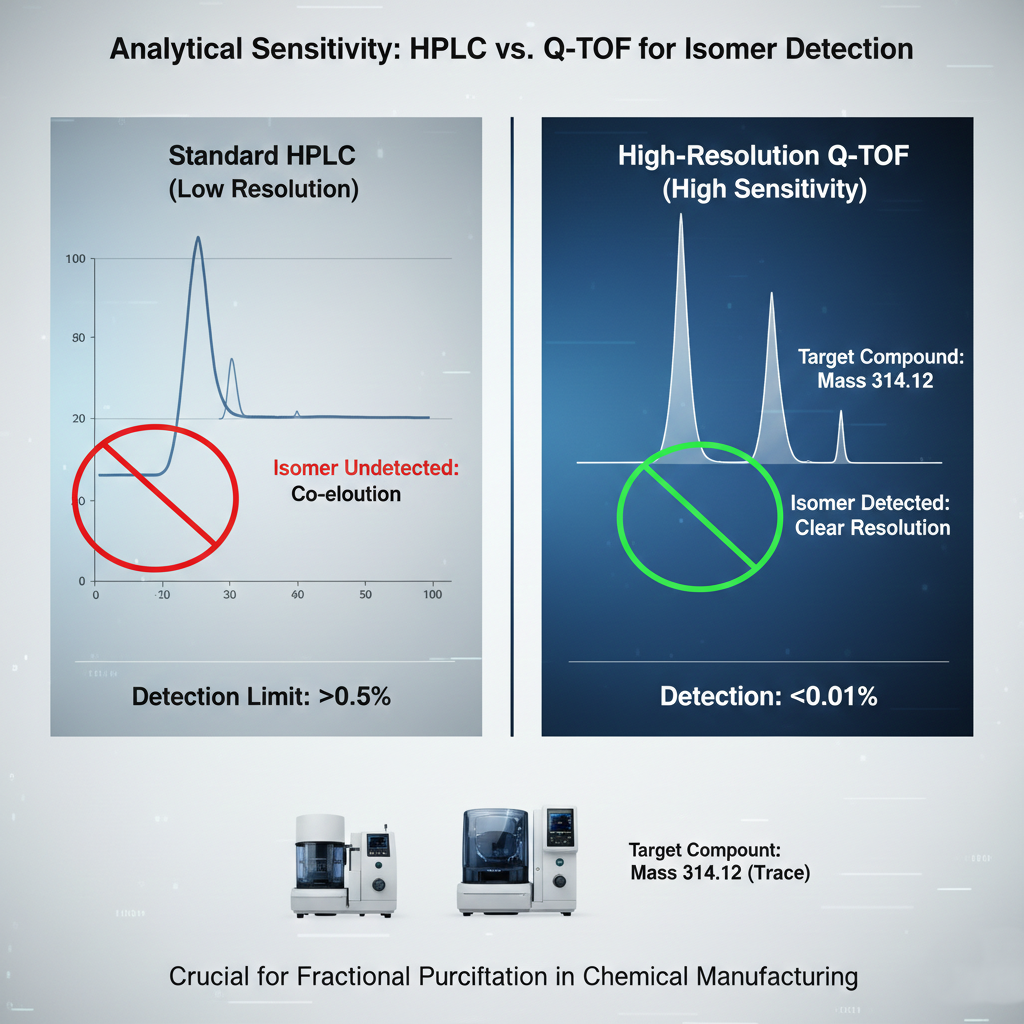
The Strategic Role of Q-TOF in Industrial Throughput
Investing in expensive, high-fidelity instrumentation like Q-TOF is a strategic decision rather than an aesthetic one. In a professional production environment, these tools provide the necessary evidence to satisfy regulatory requirements and maintain a competitive edge. The real value of high-resolution analytics lies in its ability to prevent the “quiet failures” of scale, such as blinded filter plates or unexpected seal degradation caused by unrecognized chemical interactions.
By identifying potential issues before the consequences become expensive, facilities can optimize their fractional purification in chemical manufacturing workflows for maximum efficiency. This proactive approach to data management ensures that the plant material is processed cleanly and that the real value—the high-purity filtrate—is recovered without compromise. Ultimately, high-resolution analytics transform process chemistry from an exercise in trial and error into a disciplined, data-driven science.
Conclusion
The evolution of industrial chemistry is a journey from the reactor to the data set. Mastering fractional purification in chemical manufacturing requires a commitment to analytical excellence that matches the rigor of the synthetic process. By utilizing advanced tools like Q-TOF mass spectrometry and automated recovery infrastructure, industry leaders can eliminate guesswork and achieve unprecedented levels of purity and consistency. In the world of high-volume production, the data you see is the only reality you can control.





