Introduction
Crystallization is often presented as a predictable and elegant technique within chemical education and early laboratory work. When performed at small scale, it can appear straightforward, visually satisfying, and easy to control. Clear solutions become solids, impurities are reduced, and the process seems to confirm that upstream chemistry is sound. This apparent simplicity can be misleading.
In practical process development and manufacturing, crystallization rarely fails because it is inherently unreliable. It fails because it is treated as a routine step rather than a critical test of process quality. When scale increases, crystallization becomes less forgiving and begins to reveal weaknesses that were previously ignored. Understanding crystallization as a diagnostic tool rather than a cosmetic purification step is essential for building processes that can truly scale.
Why Crystallization Appears Elegant at Small Scale
At laboratory scale, crystallization benefits from generous control. Solvent volumes are flexible, cooling rates are easily adjusted, and impurities are often present at low concentrations. Under these conditions, solids form readily and separation feels intuitive. When fines or irregular solids appear, they are often dismissed as minor inconveniences rather than meaningful signals.
This environment encourages oversimplification. Chemists may assume that solvent selection is adequate without extensive stress testing or that impurity profiles will remain manageable. The result is a process that appears functional but has not been challenged in ways that matter for larger systems.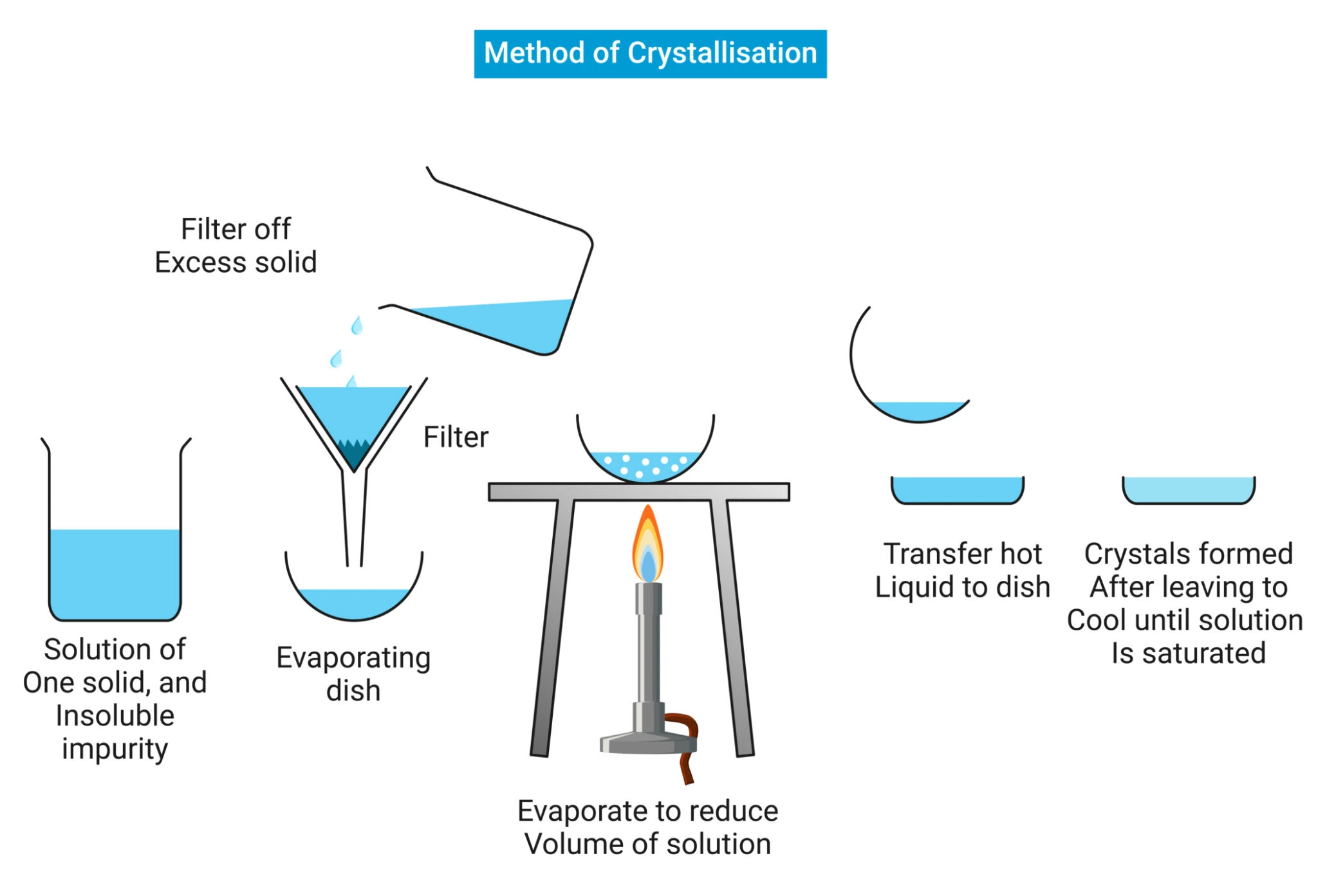
What Changes When Crystallization Is Scaled
As scale increases, crystallization begins to reflect the true health of the process. Solvent choice determines not only solubility but also viscosity and heat removal. Cooling profiles that were harmless at small volumes can generate excessive nucleation or poor crystal growth when thermal control becomes limited. Impurities that seemed insignificant can interfere with crystal formation, leading to fines, agglomeration, or filtration problems.
These outcomes are often described as unpredictability, but they are better understood as feedback. Crystallization responds directly to upstream decisions. When it produces problematic solids, it is signaling that assumptions about solvent behavior, impurity tolerance, or process robustness were incorrect.
Fines and Irregular Solids as Valuable Information
Poor crystal morphology is frequently viewed as failure. In reality, it is data. Fines indicate excessive nucleation or inadequate growth conditions. Broad particle size distributions suggest uncontrolled supersaturation or mixing limitations. These observations provide insight into how the process behaves under stress.
Rather than attempting to force crystallization to look ideal, experienced practitioners use these signals to refine upstream chemistry. Adjusting reaction conditions, improving impurity control, or redefining solvent systems often yields more benefit than repeatedly reworking the crystallization step itself.
Crystallization as a Test of Process Readiness
Crystallization should not be viewed solely as a purification tool. It is a practical evaluation of whether a process is ready to move forward. A system that crystallizes cleanly under realistic constraints is more likely to succeed in production. One that requires excessive manipulation to behave acceptably is signaling deeper issues.
By treating crystallization as a diagnostic step, chemists can identify weaknesses early, when changes are still feasible and affordable. This mindset shifts the goal from making crystals look appealing to ensuring that the overall process is resilient.
Conclusion
Crystallization does not fail because it lacks predictability. It fails when it is oversimplified and disconnected from upstream decisions. At scale, it exposes every shortcut taken earlier in development and highlights assumptions that no longer hold.
Viewing crystallization as a diagnostic rather than a decorative operation allows chemists to build stronger processes. When irregular solids appear, they should be examined, not ignored. They are evidence that the process is communicating valuable information. Listening to that information is what determines whether a process truly deserves to scale.





