Throughput Optimization: Solving the Engineering Challenges of Chemical Scaling
The transition of a chemical process from a one liter laboratory flask to a fifty liter industrial reactor represents far more than a simple increase in volume. At the benchtop scale, the primary focus remains on the kinetic and thermodynamic variables of the reaction. However, as the volume increases, the physical and mechanical limitations of the hardware begin to dictate the success of the operation. This shift marks the point where throughput ceases to be a purely chemical question and evolves into an complex engineering challenge. In high volume production environments, the reliability of seals, the durability of pumps, and the structural integrity of glassware become the primary variables that define operational uptime. Understanding these mechanical failure points is essential for any research team moving toward industrial scale manufacturing.
Mechanical Failure Modes in Large Scale Reactor Systems
When a reaction is scaled to fifty liters or beyond, the mechanical stresses on the equipment increase exponentially. Small scale laboratory setups often mask the subtle weaknesses of auxiliary components, but these issues become catastrophic bottlenecks at higher volumes. For instance, vacuum pumps that operate flawlessly at low throughput may suffer from thermal fatigue or oil contamination when forced to maintain a consistent environment for larger batches. Similarly, mechanical seals that appear robust in small glassware often experience “creep” or structural deformation under the constant pressure and chemical exposure required for industrial throughput.
Furthermore, the thermal inertia of a large scale system introduces significant risks to the physical hardware. Large borosilicate glass vessels are susceptible to thermal shock and mechanical stress, particularly during the late hours of continuous operation when temperature fluctuations are most common. These failure points are not related to the chemical reaction itself but are inherent to the physics of the container. To mitigate these risks, industrial operations must transition toward high fidelity equipment that features automated thermal monitoring and reinforced sealing mechanisms.
The Impact of Physical Variables on Molecular Stability
Scaling a reaction does not inherently change the molecular path, but it does change the environment in which that path occurs. In larger volumes, heat transfer becomes less efficient, often leading to localized “hot spots” that can degrade sensitive molecules. For researchers utilizing in silico tools to predict structure activity relationships, these physical anomalies must be accounted for to ensure the final product maintains its intended efficacy.
Predictive modeling allows scientists to visualize how a molecule might respond to the prolonged residence times and varied temperature gradients found in fifty liter systems. If a compound is categorized as “labile” or “mod labile,” any mechanical failure in the cooling or stirring system can lead to immediate molecular degradation. By integrating metabolic landscape data with engineering parameters, teams can design more resilient processes that protect the binding affinity of the target compound even when mechanical challenges arise.
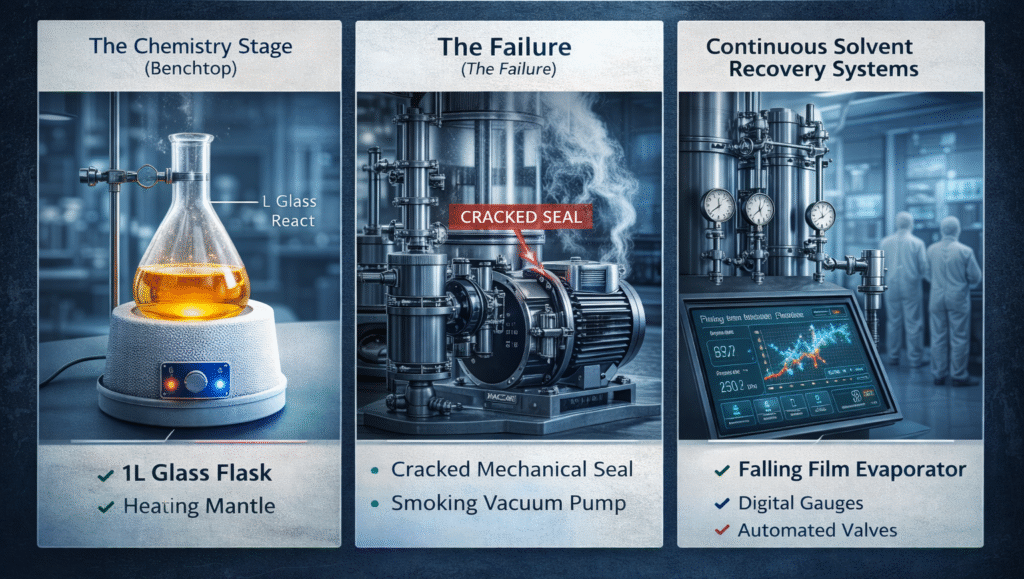
Strategic Infrastructure for Continuous Solvent Recovery Systems
One of the most effective ways to solve the engineering problems associated with scale is the implementation of continuous solvent recovery systems. Unlike traditional batch setups that rely on discrete glassware cycles, continuous solvent recovery systems are designed to handle high throughput with minimal manual intervention. These systems use automated controls to manage the pressure and flow rates that often cause mechanical failure in smaller, non-integrated setups.
The adoption of continuous solvent recovery systems reduces the stress on seals and pumps by maintaining a steady state operation rather than the repetitive cycles of heating and cooling found in batch processing. This automation allows for real time monitoring of vacuum levels and temperature, providing an early warning system for the mechanical creep or seal failure that often occurs during extended production runs. By treating throughput as an engineering objective, facilities can move away from the “trust-based” monitoring of glassware and toward a data driven, automated production environment.
Conclusion
The journey from a one liter proof of concept to a fifty liter production run is a transformative experience for any medicinal chemist. It serves as a stark reminder that while chemistry provides the blueprint, engineering provides the foundation for success. Throughput is indeed an engineering problem disguised as chemistry, and solving it requires a proactive approach to equipment selection and maintenance. By recognizing the mechanical failure modes of seals and pumps and utilizing continuous solvent recovery systems, research teams can ensure that their innovations reach the market without being sidelined by hardware limitations. As we continue to refine our chemical processes, the integration of robust engineering principles will remain the key to unlocking true industrial potential.





