Introduction
In organic chemistry, purification choices often default to what feels fastest and most familiar. Many laboratories instinctively turn to chromatographic techniques when faced with complex reaction mixtures. While these methods are powerful, they are not always the most effective or sustainable option. Crystallization remains one of the most underappreciated purification approaches, despite its deep scientific foundation and strong advantages at scale.
Crystallization is not simply a way to obtain solids. It is a selective process governed by thermodynamics and molecular behavior. When applied with understanding, it can deliver high purity material with lower cost and fewer downstream complications. Recognizing its value requires shifting perspective from speed to strategy.
Why Chromatography Became the Default Choice
Chromatography dominates many research environments because it is predictable and quick to deploy. Conditions can be adjusted rapidly, separations are visually intuitive, and results appear immediate. For early discovery work, these advantages are real.
However, chromatography relies on continuous intervention. Solvent consumption is high, loading capacity is limited, and scalability introduces cost and operational burden. These limitations are often accepted because the method feels controllable. What is less appreciated is that chromatography separates by interaction strength rather than intrinsic stability, which can make it fragile as complexity increases.
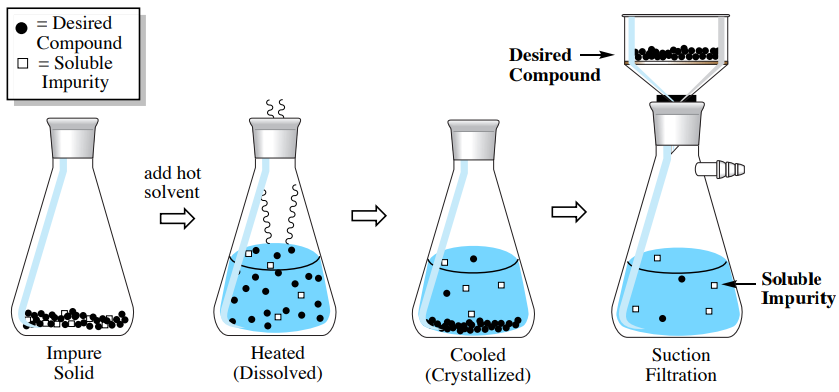
How Crystallization Enforces Selectivity Through Thermodynamics
Crystallization operates on a fundamentally different principle. Instead of forcing separation through a stationary phase, it allows molecules to organize based on energetic favorability. The desired compound enters a solid phase because it is thermodynamically preferred under specific conditions. Impurities remain in solution if they are less stable in the crystal lattice.
This behavior creates natural selectivity. When conditions are chosen correctly, crystallization excludes impurities without constant adjustment. The result is a purification step that aligns with molecular properties rather than fighting them. This is why crystallization often delivers cleaner material with fewer manipulations.
Advantages of Crystallization at Scale
When crystallization succeeds, it offers clear benefits for larger operations. Solvent use is typically lower than chromatographic methods. Equipment requirements are simpler. Solids obtained through crystallization often filter and dry more easily, reducing downstream processing effort.
Crystallization is also more tolerant of variability. Small fluctuations in composition or temperature often have limited impact when the system is well designed. This forgiving nature makes it attractive for manufacturing environments where absolute control is difficult to maintain.
The Real Challenge Is Knowing When and How to Crystallize
The difficulty with crystallization is not feasibility but judgment. Not every compound will crystallize easily, and not every system should be forced to do so. Success depends on understanding solubility behavior, solvent interactions, impurity profiles, and thermal conditions.
Effective crystallization requires patience and insight rather than pressure. Adjusting conditions to guide molecules toward their most stable form is more effective than aggressive intervention. When chemists allow the system to work with thermodynamic principles, crystallization becomes a powerful ally rather than an obstacle.
Conclusion
Crystallization remains one of the most valuable purification tools in organic chemistry when applied with understanding. It offers selectivity rooted in thermodynamics, strong scalability, and reliable downstream performance. While chromatography has its place, it should not overshadow a method that rewards insight over force.
By treating crystallization as a strategic process rather than a last resort, chemists can achieve purification that is efficient, economical, and robust. The true power of crystallization lies not in making solids form, but in knowing when to let nature do the work.





