Navigating the Three Dimensional Landscape of Chemical Architecture
Stereochemistry is the specialized branch of chemical science dedicated to investigating the three dimensional spatial arrangement of atoms within a molecule. This field of study is fundamental because the physical orientation of a molecular structure often dictates its chemical properties, reactivity, and biological behavior. At the center of this discipline is the concept of stereoisomers: molecules that share an identical molecular formula and the same connectivity of atoms but differ entirely in how those atoms are positioned in space. Understanding these subtle geometric differences is not merely a theoretical exercise; it is a critical requirement in pharmaceutical development and biochemistry, where a minor shift in atomic placement can determine whether a compound is a life-saving therapeutic or a biologically inactive substance.
The Nature of Enantiomers and Optical Activity
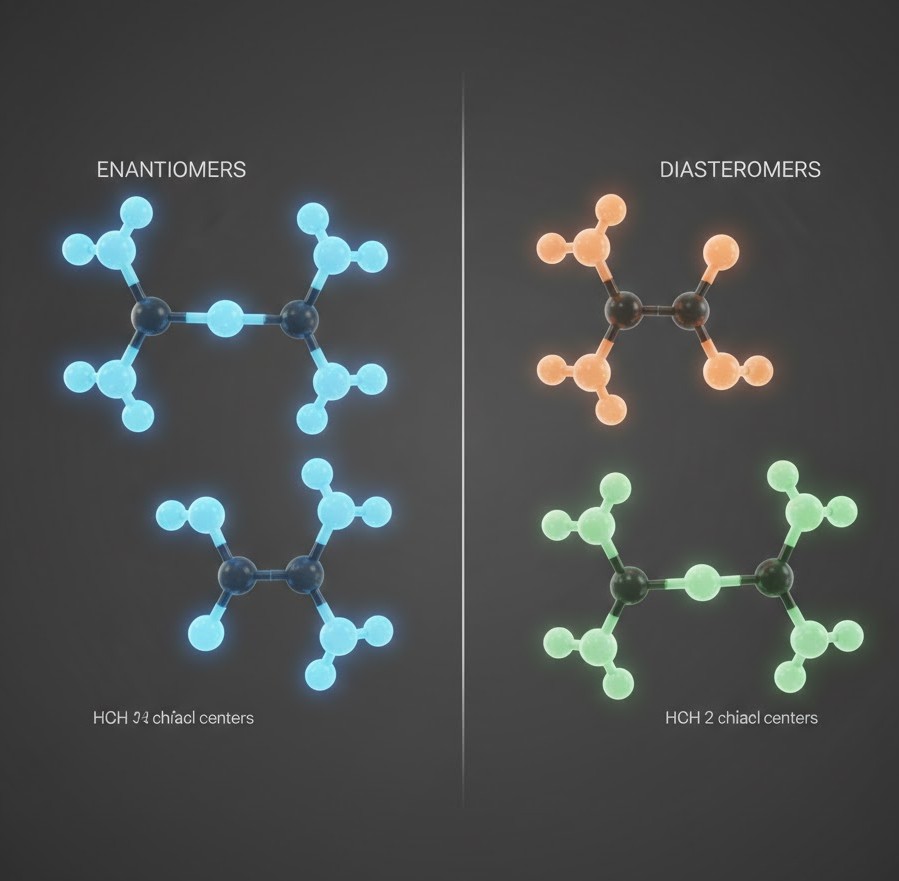
Enantiomers represent a specific class of stereoisomers characterized by a mirror image relationship. These molecules are non-superimposable, meaning that no matter how they are rotated, they cannot be perfectly aligned with their mirror counterpart, much like a person’s left and right hands. In a laboratory setting, enantiomers are particularly fascinating because they possess identical physical properties, such as boiling points, melting points, and solubilities.
The primary physical distinction between a pair of enantiomers is their optical activity. When exposed to plane polarized light, one enantiomer will rotate the light in a clockwise direction, designated as the (+) or dextrorotatory form, while the other will rotate it counterclockwise, known as the (-) or levorotatory form. While they appear identical in a test tube, their interactions within the human body can be vastly different. Biological systems are themselves chiral, meaning they are “handed,” and receptors often recognize only one specific enantiomer, leaving the other either ineffective or potentially harmful.
Structural Divergence in Diastereomers
Unlike enantiomers, diastereomers are stereoisomers that do not share a mirror image relationship. These compounds typically arise in molecules that possess two or more chiral centers. When the configuration at one or more of these centers is changed while others remain the same, the resulting molecules are diastereomers. Because they are not mirror images, they do not possess the same physical or chemical properties.
Diastereomers often exhibit different melting points, boiling points, and chromatographic retention times. This physical divergence makes them significantly easier to separate in a laboratory environment compared to enantiomers. A classic illustration of this concept is found in the cis-trans isomerism of alkenes, where the relative positioning of functional groups across a double bond creates distinct molecules with unique stability and reactivity profiles. In drug design, the creation of specific diastereomers allows chemists to fine tune the physical characteristics of a drug to improve its solubility or absorption.
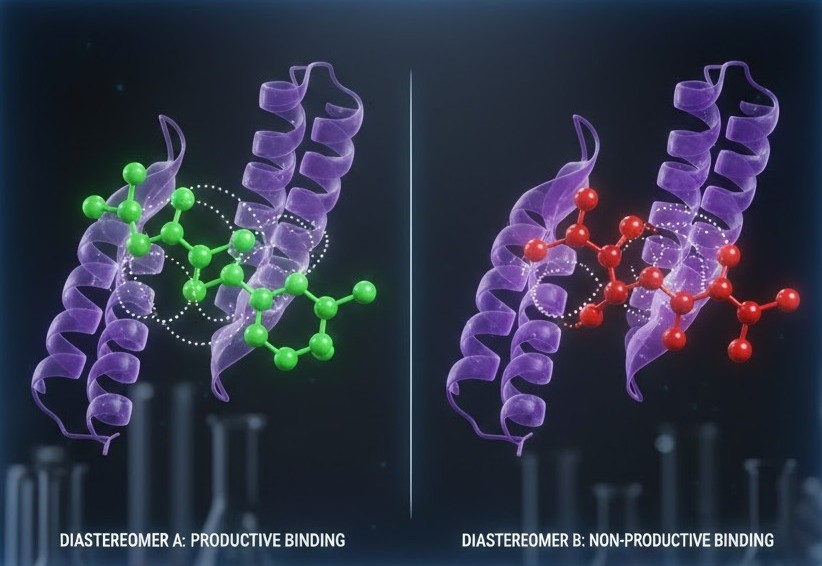
Pharmacological Implications and Molecular Precision
The distinction between enantiomers and diastereomers is of paramount importance in the fields of pharmacology and medicinal chemistry. Most drugs function by binding to specific protein receptors or enzymes, which are highly sensitive to molecular geometry. If a molecule’s three dimensional shape is not an exact match for the target site, its efficacy is lost. This “lock and key” mechanism means that developing a pure stereoisomer is often the difference between a successful treatment and a failed clinical trial.
By mastering the study of diastereomers, researchers gain the ability to predict how a molecule will behave in a complex biological environment. This precision allows for the development of more effective medications with fewer off-target effects. As we continue to push the boundaries of synthetic chemistry, the ability to control and analyze the spatial arrangement of atoms remains the gold standard for creating the next generation of materials and therapeutics.
Conclusion
Stereochemistry provides the essential framework for understanding how the three dimensional arrangement of atoms impacts the macroscopic world. Whether through the mirror image relationship of enantiomers or the distinct physical properties of diastereomers, the geometry of a molecule is inseparable from its function. For scientists and industry experts, the study of these isomers is a foundational tool for innovation, ensuring that every chemical reaction and biological process is understood at the most granular, atomic level.