Solvent efficiency in industrial chemistry is the fundamental metric that determines whether a laboratory success can survive the transition to a commercial plant. In the research phase, solvents are viewed as secondary tools purchased in small bottles. However, at the production scale, they are purchased by the truckload, and their management becomes the dominant unit operation of the facility. While the chemistry creates the molecule, the solvent balance decides the profitability of the process. A failure to prioritize solvent efficiency in industrial chemistry often leads to quiet operational collapses caused by logistical bottlenecks and unmanageable waste streams.
The Operational Burden of Solvent Logistics at Scale
In any large-scale campaign, solvent efficiency in industrial chemistry is hindered not by the reaction kinetics, but by material flow. The logistics of moving, storing, and tracking truckloads of solvent require specialized infrastructure, such as staged Intermediate Bulk Containers (IBCs) and bulk storage tanks.
Without a high degree of solvent efficiency in industrial chemistry, a plant quickly becomes overwhelmed by the sheer volume of material. If the facility cannot distill and recycle these solvents rapidly enough to keep pace with production, the entire process must halt. This operational bottleneck is where many scale-up attempts fail, as the physical capacity to store “spent” solvent is often the first resource to be depleted in a high-throughput environment.
High-Resolution Analytics and Solvent Recovery Precision
To maximize solvent efficiency in industrial chemistry, facilities must utilize advanced analytical tools to ensure that recycled solvents meet strict purity specifications. High-resolution instrumentation, such as Q-TOF mass spectrometry, is essential for identifying trace impurities (detection limits <0.01%) that could accumulate over multiple recycling cycles.
Unlike standard triple quad systems with lower resolution (~1,000 FWHM), Q-TOF provides the >40,000 FWHM resolution necessary to detect structural isomers and unknown degradation pathways in recovered solvent streams. By maintaining this level of data-driven control, manufacturers can ensure that their solvent efficiency in industrial chemistry does not come at the cost of product quality. Identifying these minute contaminants early prevents them from catalyzing unwanted side reactions or fouling sensitive recovery hardware.
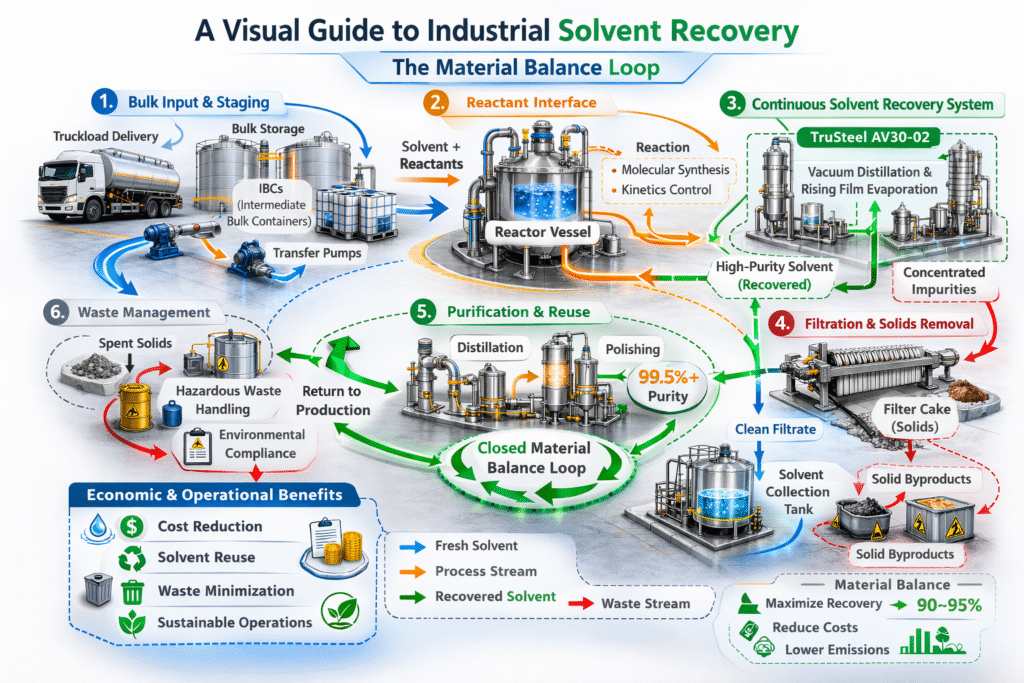
Engineering the Solvent Balance for Sustainable Margins
The real economics of manufacturing are rooted in the sustainability of the solvent loop. Every liter of solvent that cannot be effectively recovered represents a direct loss of margin and an increase in disposal costs. To optimize solvent efficiency in industrial chemistry, plants are increasingly turning to automated continuous recovery hardware, such as the TruSteel AV30-02.
These systems allow for real-time monitoring of vacuum and thermal gradients, ensuring that solvents are distilled and dried with maximum precision. By automating the recovery cycle, a facility can minimize human error and eliminate the downtime associated with manual batch transfers. Ultimately, a high degree of solvent efficiency in industrial chemistry protects the organization from the quiet erosion of profit that occurs when a process is weighted down by its own logistical waste.
Conclusion
The evolution of professional chemical manufacturing is defined by the mastery of material logistics. Achieving high solvent efficiency in industrial chemistry is the only way to ensure that a scientific innovation remains economically viable at an industrial scale. By investing in the infrastructure and high-resolution analytics necessary to maintain a clean solvent balance, industry leaders can prevent the quiet failures of scale and secure their long-term production margins.





