Navigating the Synthetic and Biological Frontier of Cannabinoid Development
The shift from botanical extraction to sophisticated drug design in the cannabinoid industry has introduced a critical need for rigorous safety protocols. As researchers develop novel chemical entities and semi-synthetic analogs, the primary challenge remains ensuring these compounds are safe for human consumption across diverse delivery methods. Safety evaluation is no longer limited to simple purity checks; it now encompasses a multidisciplinary approach combining predictive computational modeling with high-precision experimental validation. This holistic framework is essential for mitigating the risks of off-target toxicity and cardiovascular events, ultimately bridging the gap between innovative research and consumer protection.
Predictive Pharmacokinetics: The Power of In Silico ADME Modeling
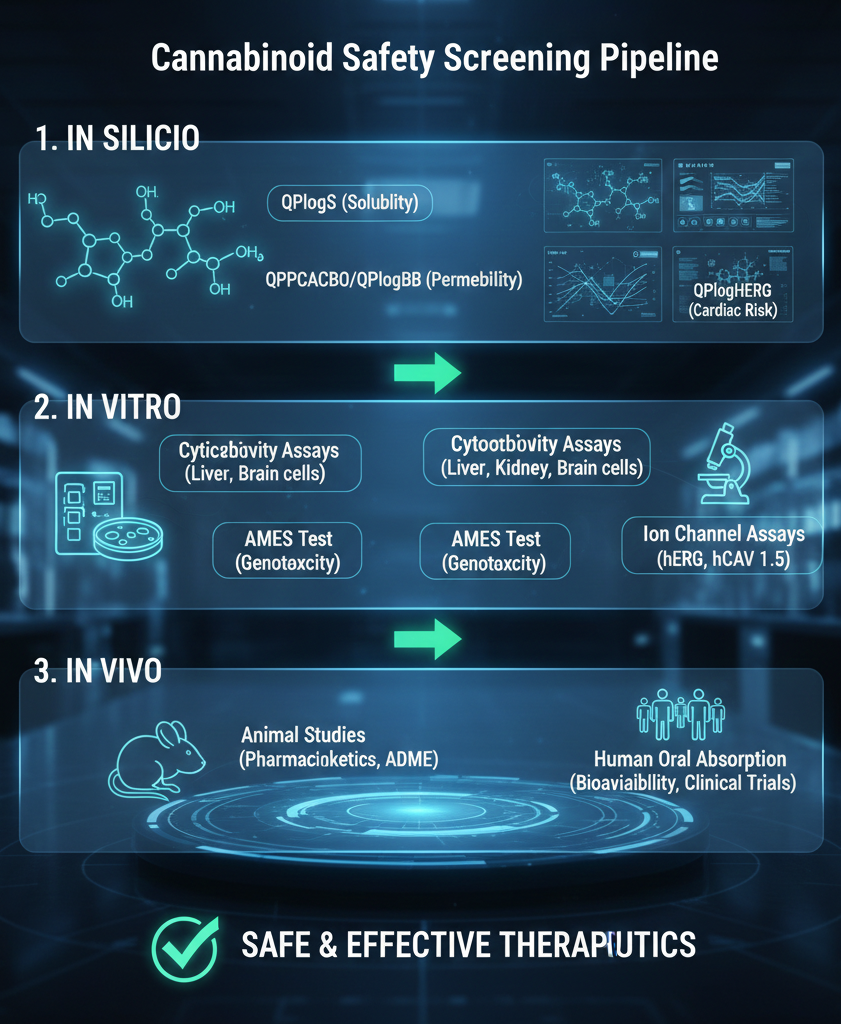
At the forefront of modern cannabinoid research is In Silico ADME modeling, which utilizes advanced computational algorithms to predict how a compound will interact with the human body. By simulating Absorption, Distribution, Metabolism, and Excretion (ADME) properties, researchers can identify potential pharmacological failures long before entering the laboratory. This digital screening process is crucial for assessing bioavailability, as many cannabinoids exhibit high lipophilicity that can severely limit their oral absorption.
Key computational metrics such as QPlogS are used to assess a molecule’s aqueous solubility. Because many cannabinoids are naturally insoluble in water, understanding their ability to dissolve in biological fluids is vital for determining effective dosing. Furthermore, models like QPPCACO and QPlogBB evaluate the compound’s ability to cross critical biological barriers, such as the gut-blood barrier and the blood-brain barrier (BBB). Predicting BBB penetration is particularly significant for cannabinoids, as it determines whether a compound will have psychoactive or therapeutic effects within the central nervous system.
Cardiovascular and Genotoxic Safety: Screening Beyond Potency
Cardiovascular safety is a paramount concern, specifically regarding the inhibition of the human ether-a-go-go related gene (hERG). The QPlogHERG model and subsequent ion channel assays identify whether a compound might block potassium channels in the heart, a phenomenon known to cause cardiac arrhythmias and QT interval prolongation. Early screening for these interactions is mandatory for regulatory compliance and ensures that new cannabinoid therapeutics do not pose a hidden risk to heart health.
Beyond cardiac safety, the AMES test serves as the gold standard for evaluating genotoxicity. This bacterial assay screens for mutagenic potential, identifying substances that could cause DNA damage or lead to carcinogenicity. Given the long-term nature of many cannabinoid treatments, ensuring that a compound does not have mutagenic properties is a non-negotiable step in the safety profile. These predictive and experimental tools provide a comprehensive defense against the unintended long-term biological consequences of novel compounds.
Experimental Validation: Cytotoxicity and Ion Channel Assays
While computational models provide a roadmap, rigorous experimental assessments are necessary to confirm organ-specific safety. Cytotoxicity testing evaluates the impact of cannabinoids on vital organs, including the liver, kidneys, and lungs. For instance, studies have shown that high concentrations of certain cannabinoids can induce significant cellular damage in human hepatic cells, highlighting the importance of defining safe therapeutic windows.
Comprehensive safety evaluation also involves specialized ion channel assays, such as hCAV 1.5 and hNAV1.2, which assess cardiac and neuronal stability beyond the hERG channel. These assays identify potential cardiotoxic and neurotoxic concerns by measuring how a compound affects the electrical activity of cells. By integrating these experimental findings with in silico data, cannabis manufacturers can provide a robust, evidence-based safety dossier that satisfies both regulatory bodies and increasingly health-conscious consumers.
Conclusion
The evolution of cannabinoid safety testing marks a significant step toward the “pharmaceuticalization” of the industry. By leveraging In Silico ADME modeling alongside rigorous AMES and cytotoxicity testing, researchers can develop compounds that are not only effective but also biologically compatible. As consumers become more aware of the complexities of cannabinoid pharmacology, the demand for this level of transparent, high-quality data will continue to grow. Manufacturers who embrace these comprehensive safety approaches are not just complying with regulations—they are leading the industry toward a future of innovation grounded in scientific integrity and public trust.





