The rapidly maturing cannabinoid industry demands a transition from basic compliance testing to sophisticated analytical characterization. Among the suite of tools available to the medicinal chemist, Nuclear Magnetic Resonance (NMR) spectroscopy stands as the gold standard for verifying molecular identity and assessing structural purity. Unlike standard chromatographic methods that may fail to resolve closely related isomers, NMR provides a detailed electronic signature of every proton environment within a molecule. This capability is particularly critical during the synthesis of minor cannabinoids, where subtle variations in reaction conditions can lead to the formation of structurally similar contaminants. By leveraging high-field NMR, researchers can achieve a level of transparency that ensures therapeutic consistency and safety across clinical and industrial applications.
Identifying Co-Synthesized Contaminants in CBC Production
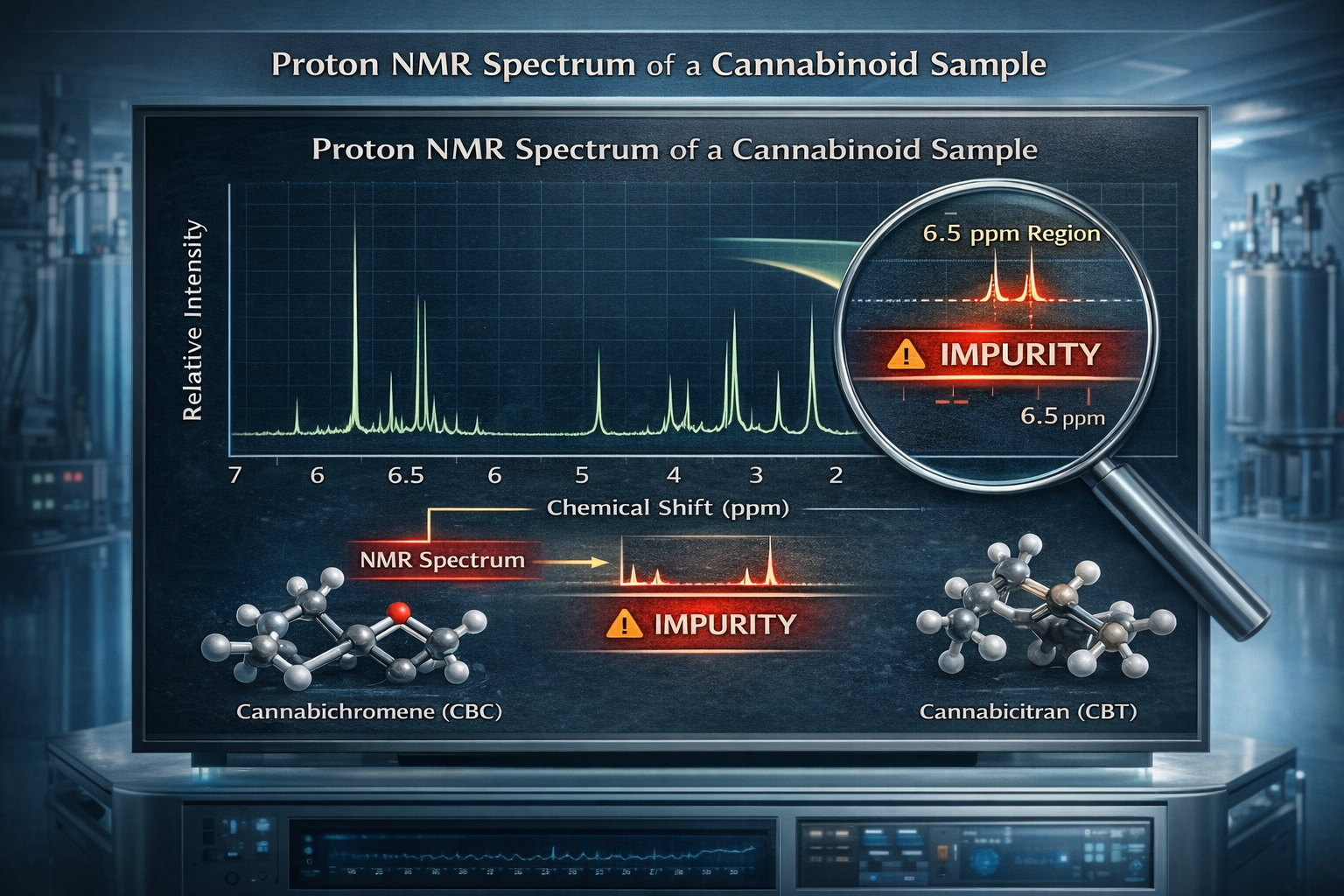
In the synthesis of Cannabichromene (CBC), the presence of Cannabicitran (CBT) as a co-synthesized byproduct is a frequent analytical challenge. Because these two molecules often emerge from the same synthetic pathways, traditional purification techniques may leave trace amounts of CBT within the CBC sample. Proton NMR allows for the direct visualization of this contamination by revealing peaks that do not belong to the CBC backbone. While the major CBC protons manifest as tall, well-defined signals, the presence of CBT is betrayed by distinctive secondary peaks.
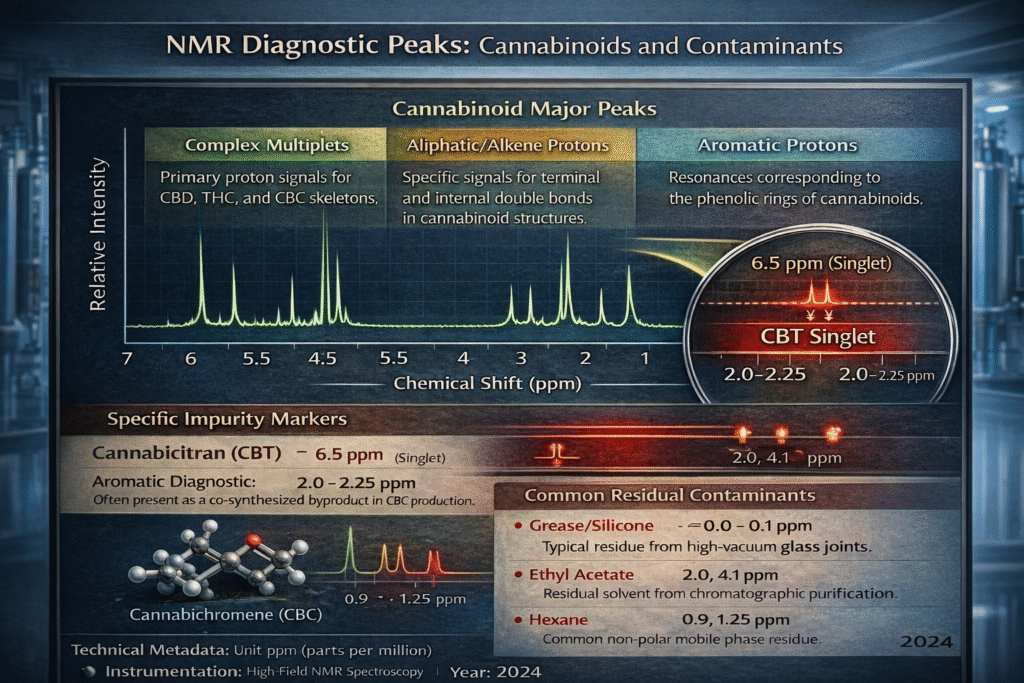
Specifically, the appearance of small singlets in the region of 6.5 parts per million (ppm) serves as a diagnostic indicator for the aromatic protons of CBT. Further confirmation can be found in the aliphatic region, where additional signals appearing around 2.0 ppm and 2.25 ppm indicate the specific proton environments of the CBT contaminant. By quantifying the integration of these minor signals against the primary peaks, chemists can determine the exact percentage of impurity, a step that is vital for refining purification protocols and meeting stringent pharmaceutical specifications.
Detection of Residual Solvents and Laboratory Artifacts
Beyond the identification of synthetic byproducts, NMR is an unrivaled tool for detecting “invisible” laboratory contaminants that might pass through standard screening. During the final stages of purification, chromatography often employs volatile organic compounds such as hexane and ethyl acetate. While rotary evaporation is intended to remove these substances, residual traces can remain trapped within the cannabinoid matrix. NMR spectroscopy identifies these common culprits with extreme precision, as hexane and ethyl acetate possess highly characteristic chemical shifts that do not overlap with the complex multiplet structures of the cannabinoid skeleton.
Furthermore, NMR can detect non-chemical artifacts such as high-vacuum grease used on glass joints during the distillation or synthesis process. Grease typically appears as a sharp, intense singlet in the upfield region of the spectrum, providing immediate evidence of a breach in laboratory cleanliness or handling protocols. Identifying these residuals early in the development cycle is essential for maintaining a “green” and safe manufacturing process, as well as avoiding the inhibitory effects that certain solvents can have on downstream biological assays or formulation stability.
Strategic Implications for Quality Control and Formulation
The ability to perform deep-dive impurity profiling via NMR has profound implications for the commercial viability of a cannabinoid project. High-purity samples are a prerequisite for establishing accurate Structure-Activity Relationships (SAR) and determining the true potency of a novel scaffold. If a researcher assumes a sample is 100% CBC while it actually contains 5% CBT, the resulting biological data will be skewed, potentially leading to incorrect conclusions about binding affinity or cellular efficacy.
Implementing NMR as a standard quality control gate ensures that only the most refined compounds proceed to in-vitro or in-vivo testing. This proactive approach to analytical chemistry reduces the risk of late-stage failures and provides a robust data package for regulatory filings. For industry experts, the mastery of NMR interpretation is not merely an academic exercise; it is a critical business function that protects the integrity of the research and the safety of the end consumer.
Conclusion
Nuclear Magnetic Resonance spectroscopy is the cornerstone of structural integrity in the cannabinoid space. From identifying specific synthetic contaminants like CBT in CBC samples to detecting minute traces of residual solvents and laboratory grease, NMR provides a comprehensive view of a compound’s purity profile. As the industry continues to evolve toward pharmaceutical standards, the reliance on high-resolution analytical data will only increase. By integrating NMR into the standard development workflow, researchers can ensure that their cannabinoid oncology and therapeutic projects are built on a foundation of absolute molecular clarity, paving the way for the next generation of safe and effective cannabinoid-based medicines.