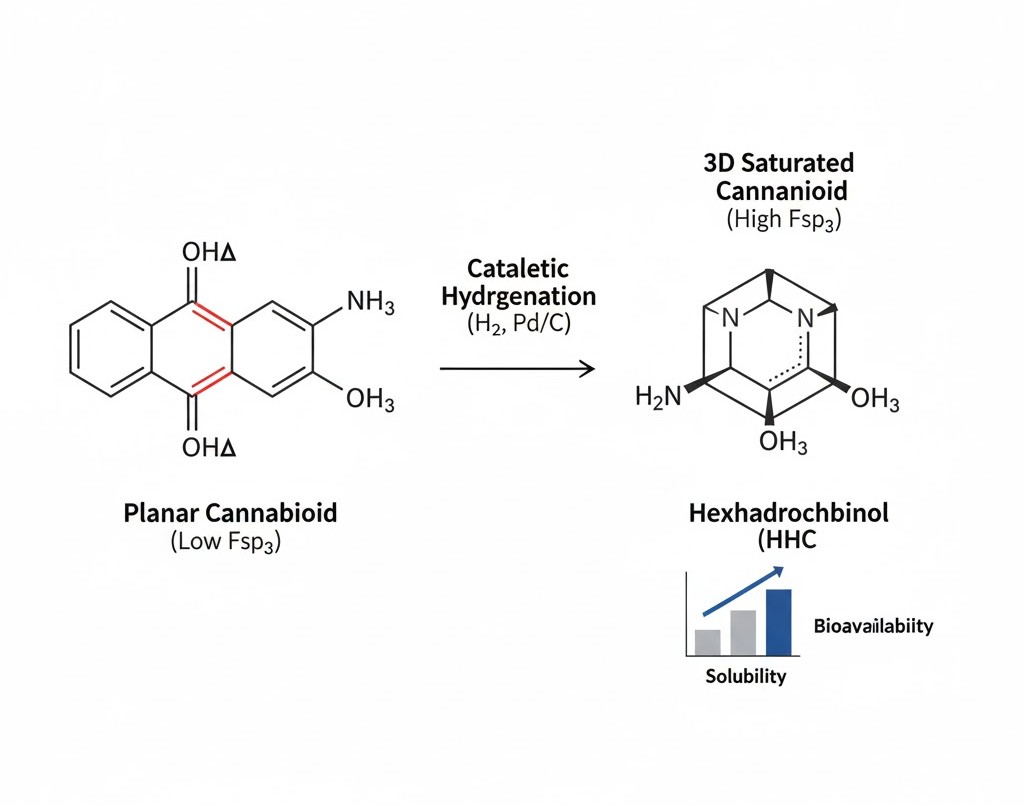
Beyond the Planar Molecule: Engineering Three-Dimensionality for Enhanced Efficacy
The transition of the cannabis industry from simple botanical extraction to rigorous pharmaceutical science has highlighted the necessity for advanced molecular modification. Hydrogenation, a fundamental reaction in organic chemistry involving the addition of molecular hydrogen to unsaturated compounds, is at the forefront of this evolution. By strategically altering the chemical architecture of cannabinoids, researchers can overcome inherent limitations such as oxidative instability and poor pharmacokinetic profiles. This process does not merely change the physical state of the compound; it fundamentally redefines its interaction with the human endocannabinoid system, paving the way for a new generation of high-purity, long-lasting therapeutic agents.
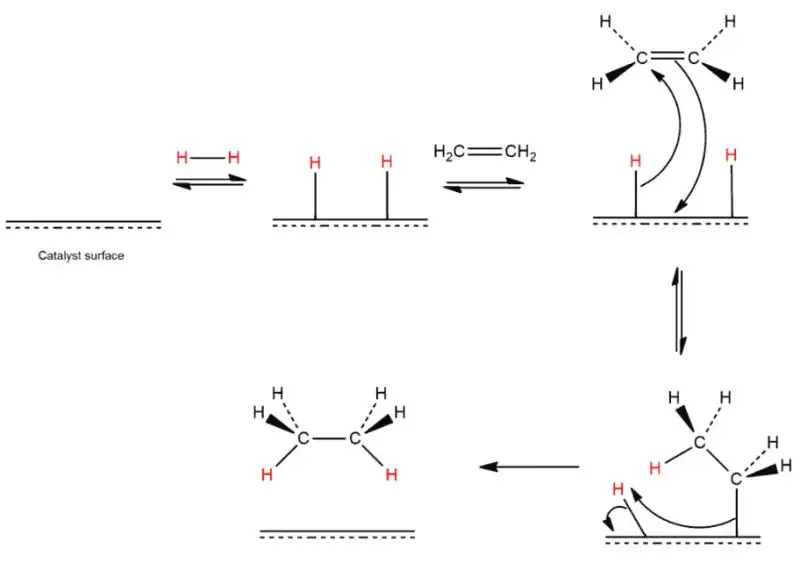
Enhancing Oxidative Stability and Product Longevity
Naturally occurring cannabinoids, such as THC and CBD, possess multiple double bonds within their structure that are highly susceptible to oxidation. Exposure to light, heat, and oxygen triggers a degradation process that often converts these active compounds into less potent metabolites like Cannabinol (CBN). Hydrogenation solves this by saturating these double bonds, effectively “locking” the molecule into a more stable state.
When a cannabinoid undergoes catalytic hydrogenation, the carbon-carbon double bonds are transformed into single bonds. This shift significantly reduces the molecule’s vulnerability to thermal and oxidative stress. For manufacturers, this translates to products with an exponentially longer shelf life and more consistent dosing. A primary real-world example is the synthesis of Hexahydrocannabinol (HHC) from THC. By saturating the cyclohexene ring, the resulting HHC molecule is far more resistant to the environmental factors that typically degrade standard cannabis distillates.
The Escape from Flatland: Increasing Fsp3 Fraction
A critical concept in modern medicinal chemistry is the “Escape from Flatland,” a principle famously detailed by Frank Lovering. This concept argues that molecules with a higher fraction of $sp^3$ hybridized carbons ($Fsp^3$) tend to be more successful in clinical trials. Naturally occurring cannabinoids often possess high aromaticity and planar (flat) structures. Hydrogenation increases the $Fsp^3$ fraction by converting flat, $sp^2$ hybridized double bonds into three-dimensional, $sp^3$ hybridized single bonds.
Increasing molecular complexity and saturation leads to a more three-dimensional structure, which offers several pharmacological advantages. These “3D” molecules often exhibit improved solubility and better permeability through biological membranes. In the context of drug discovery, a higher $Fsp^3$ fraction is frequently correlated with increased target specificity. By moving away from flat molecular architectures, we reduce the likelihood of “promiscuous” binding, where a molecule interacts with unintended receptors, thereby minimizing potential off-target side effects and drug-drug interactions.
Optimizing Bioavailability and Therapeutic Reach
Bioavailability remains one of the greatest challenges in cannabinoid delivery. Because these compounds are naturally lipophilic, they often suffer from poor absorption in the aqueous environment of the human body. Hydrogenation can be used to fine-tune the lipophilicity of a cannabinoid, optimizing how it is absorbed and distributed.
Hydrogenated derivatives often show enhanced pharmacokinetic properties, allowing for higher plasma concentrations at lower doses. Furthermore, the structural changes induced by hydrogenation can alter the binding affinity for CB1 and CB2 receptors. For instance, hydrogenated CBD (H4CBD) has been studied for its potential to interact differently with the endocannabinoid system compared to its precursor. This ability to synthesize analogs with tailored properties allows scientists to develop specific treatments for conditions like chronic pain or neuroinflammation with a level of precision that raw extracts cannot provide.
Conclusion
The application of hydrogenation and the deliberate increase of the $Fsp^3$ fraction represent a sophisticated leap forward in cannabinoid research. By moving beyond the limitations of naturally occurring plant chemistry, the industry can produce molecules that are not only more stable but also more biologically compatible. As we continue to apply the principles of pharmaceutical drug discovery to the cannabis space, the focus on molecular three-dimensionality and saturation will remain the gold standard for creating safe, effective, and innovative therapeutic products.