Beyond Binding Affinity: The Importance of Ion Channel Profiling
In the contemporary era of drug discovery, the focus has shifted from mere target binding to a comprehensive understanding of a compound’s systemic safety profile. Central to this evolution is the study of ion channels, specialized membrane proteins that regulate the flow of ions across cellular membranes. Among the most critical for human safety are the hERG, hCAV 1.2, and hNAV 1.5 channels. These proteins orchestrate the delicate electrical signaling required for cardiac rhythm and neuronal excitability. Understanding their modulation is no longer optional; it is a regulatory and scientific necessity to prevent adverse drug reactions and ensure the successful transition of a lead compound from the bench to the bedside.
Cardiac Repolarization and the Sentinel Role of hERG Channels
Shutterstock
When a drug inadvertently blocks hERG channels, it delays cardiac repolarization, leading to a condition known as QT interval prolongation. This clinical marker is associated with an increased risk of Torsade de Pointes, a potentially fatal ventricular arrhythmia. Consequently, hERG screening has become a mandatory gatekeeper in the drug development pipeline. Researchers utilize patch-clamp electrophysiology or fluorescence-based assays to ensure that new chemical entities do not interfere with this vital potassium current, thereby safeguarding patients against drug-induced cardiotoxicity.
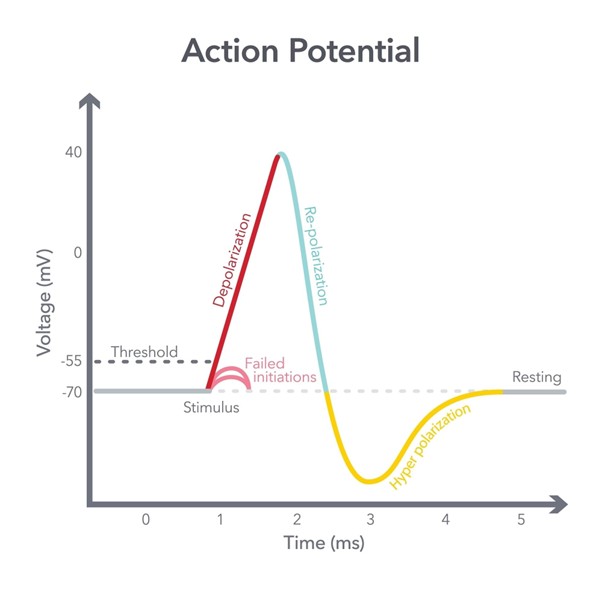
Orchestrating Cellular Dynamics via hCAV 1.2 Calcium Regulation
While potassium channels manage the end of a signal, voltage-gated calcium channels like hCAV 1.2 are responsible for translating electrical signals into physiological actions. The hCAV 1.2 channel regulates the influx of calcium ions into cardiovascular and neuronal cells, acting as a primary trigger for muscle contraction, gene expression, and the release of neurotransmitters. Because calcium is a ubiquitous secondary messenger, any off-target modulation of hCAV 1.2 can have profound systemic consequences.
In the cardiovascular system, hCAV 1.2 is the dominant calcium channel in the myocardium and vascular smooth muscle. Inhibition of this channel can lead to decreased cardiac contractility or vasodilation, whereas over-activation may cause hypertension or arrhythmias. In the central nervous system, these channels are linked to cognitive functions and mood regulation. Therefore, profiling a compound’s interaction with hCAV 1.2 is essential for maintaining hemodynamic stability and avoiding neurological side effects during therapeutic development.
hNAV 1.5 and the Propagation of Action Potentials
The hNAV 1.5 channel is a voltage-gated sodium channel that serves as the primary driver of the rapid upstroke phase of the action potential in the heart and specific neuronal tissues. By allowing a rapid influx of sodium ions, hNAV 1.5 facilitates the initiation and propagation of the electrical impulses that allow for synchronized heartbeats and rapid neuronal communication. The integrity of this channel is paramount for maintaining the conduction velocity of electrical signals through the cardiac conduction system.
Dysregulation of hNAV 1.5, whether through genetic mutation or drug-induced interference, is heavily implicated in a range of pathologies, including Brugada syndrome and various forms of epilepsy. For the drug researcher, assessing hNAV 1.5 activity is critical to ensure that a candidate molecule does not slow conduction velocity or predispose the heart to reentry arrhythmias. By characterizing the interaction between a lead compound and this sodium channel, scientists can better predict the potential for neuronal excitability issues and cardiac conduction delays.
Conclusion
The intricate dance of cellular signaling is dependent upon the precise function of ion channels. The hERG, hCAV 1.2, and hNAV 1.5 channels represent a critical triad in safety pharmacology, governing the essential rhythms of the heart and the excitability of the nervous system. As drug discovery becomes increasingly complex, the integration of electrophysiological profiling early in the development process is the only way to ensure the delivery of safe and effective therapeutics. Moving forward, the collaboration between computational modeling and experimental ion channel assays will continue to be the cornerstone of a rigorous, safety-first approach to medicine.





