From Botanical Extract to Designer Isolate: Navigating the Synthetic Frontier
The evolution of the cannabis industry from simple agricultural extraction to sophisticated pharmaceutical manufacturing has necessitated a shift toward advanced organic synthesis. At the heart of this transition is the concept of chemical stability and molecular tailoring. Hydrogenation, a cornerstone of organic chemistry, has emerged as a critical methodology for chemists seeking to modify the structural framework of cannabinoids, ensuring that products meet the rigorous standards of modern clinical research.
The Mechanics of Alkene Saturation and Stability
One of the most prominent applications of hydrogenation in the cannabis space is the saturation of alkenes and alkynes. Naturally occurring cannabinoids like Delta-9 THC and CBD contain unsaturated carbon-carbon double bonds that are highly susceptible to oxidative degradation. By employing catalytic hydrogenation—often using palladium on carbon (Pd/C) or platinum dioxide (PtO2)—chemists can break these double bonds and insert hydrogen atoms to form more stable alkanes.
A prime real-world example is the conversion of Delta-8 THC into Hexahydrocannabinol (HHC). This structural modification eliminates the double bond in the cyclohexene ring, which is the primary site for oxidation. The result is a molecule with a significantly longer shelf life and a unique binding affinity for the CB1 and CB2 receptors.
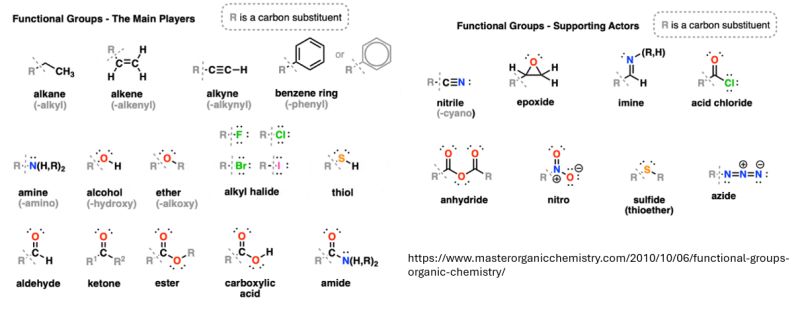
Modification of Aromatic Systems and Nitrogenous Groups
While cannabinoids are primarily terpenophenolic, the introduction or modification of aromatic rings and nitrogen-containing functional groups opens new doors for drug discovery. Aromatic rings can be partially or fully hydrogenated into cyclohexane derivatives to alter the molecule’s planar geometry. This change directly impacts the “lock and key” mechanism of endocannabinoid receptors.
Furthermore, the reduction of nitro groups or imines into amines is a vital pathway for creating semi-synthetic cannabinoid derivatives. These amine functionalities can drastically change the pharmacokinetics of a compound, potentially improving its water solubility or crossing the blood-brain barrier more efficiently. For an industry focused on targeted therapeutics, the ability to transition from a prochiral imine to a stable amine is a major strategic advantage.
Carbonyl Reduction and Carboxylic Acid Transformation
The reduction of ketones and aldehydes into alcohols is another essential tool in the cannabinoid chemist’s arsenal. Carbonyl groups are often reactive centers; by converting them into hydroxyl groups, researchers can produce more stable derivatives. For instance, modifying carbonyl-containing cannabinoids can lead to alcohol derivatives that are easier to formulate in aqueous environments.
Similarly, the reduction of carboxylic acids and esters can transform acidic cannabinoids (like THCA or CBDA) into neutral forms. While decarboxylation is the traditional route, selective hydrogenation allows for a more controlled transformation that preserves other sensitive parts of the molecular scaffold. This precision ensures that the final product maintains a high degree of purity without unwanted byproducts.
Conclusion
Hydrogenation represents a bridge between raw botanical extracts and high-efficacy medicinal agents. By mastering the reactivity of various functional groups, the industry can move beyond what the plant naturally provides, developing stable, bioavailable, and novel therapeutic agents. The future of the industry lies in the precision of the reactor and the mastery of molecular geometry.