Fractional purification in chemical manufacturing is often discussed in the context of molecular precision and yield, but the industrial reality of scaling a process centers on the transition from experimental chemistry to complex operational logistics. When a project moves beyond the laboratory, the definition of scaling shifts from simply using larger reactors to managing significantly higher consequences. In a benchtop environment, a procedural error may result in the loss of a few grams of material, but at production scale, the same mistake can lead to the loss of entire truckloads of product. Successful scaling requires a fundamental pivot toward infrastructure management, safety protocols, and waste stream logistics to ensure that the increased throughput does not lead to catastrophic operational failure.
Infrastructure and Bulk Material Management at Scale
At the production scale, the primary challenges are often found outside the reactor vessel. Real-world scaling involves the coordination of bulk storage facilities, staged Intermediate Bulk Containers (IBCs), and sophisticated logistics networks. While the chemistry remains the core of the process, the operational bottleneck often resides in the physical movement and storage of raw materials and final products. Large-scale facilities must implement rigorous safety systems and automated infrastructure to maintain a environment where “everything must work” simultaneously.
For instance, managing the sheer volume of solvents required for extraction necessitates industrial-grade solvent recovery systems. Automated hardware, such as the TruSteel AV30-02, provides the necessary control to handle high-throughput processing while minimizing human error in hazardous zones. By integrating bulk storage with automated recovery, a facility can maintain continuous production cycles that would be impossible to manage through manual benchtop methods.
The Shift from Chemical Kinetics to Operational Survivability
In the laboratory, researchers focus on proving that a reaction pathway is feasible and repeatable. However, as the process enters the production phase, the focus must shift to whether the process is “survivable” from an operational standpoint. This involves a proactive analysis of failure modes that are often invisible at the bench scale. Mechanical integrity, seal durability, and pump reliability become the defining variables of success in fractional purification in chemical manufacturing.
A key aspect of this survivability is the management of waste streams. Industrial chemistry generates significant amounts of byproducts and spent solvents that must be recovered or disposed of according to strict environmental and safety regulations. Scaling up means that these waste streams become as critical as the target product. High-capacity filtration and recovery units are used to ensure that waste is processed efficiently, preventing it from becoming a bottleneck that halts the entire production line.
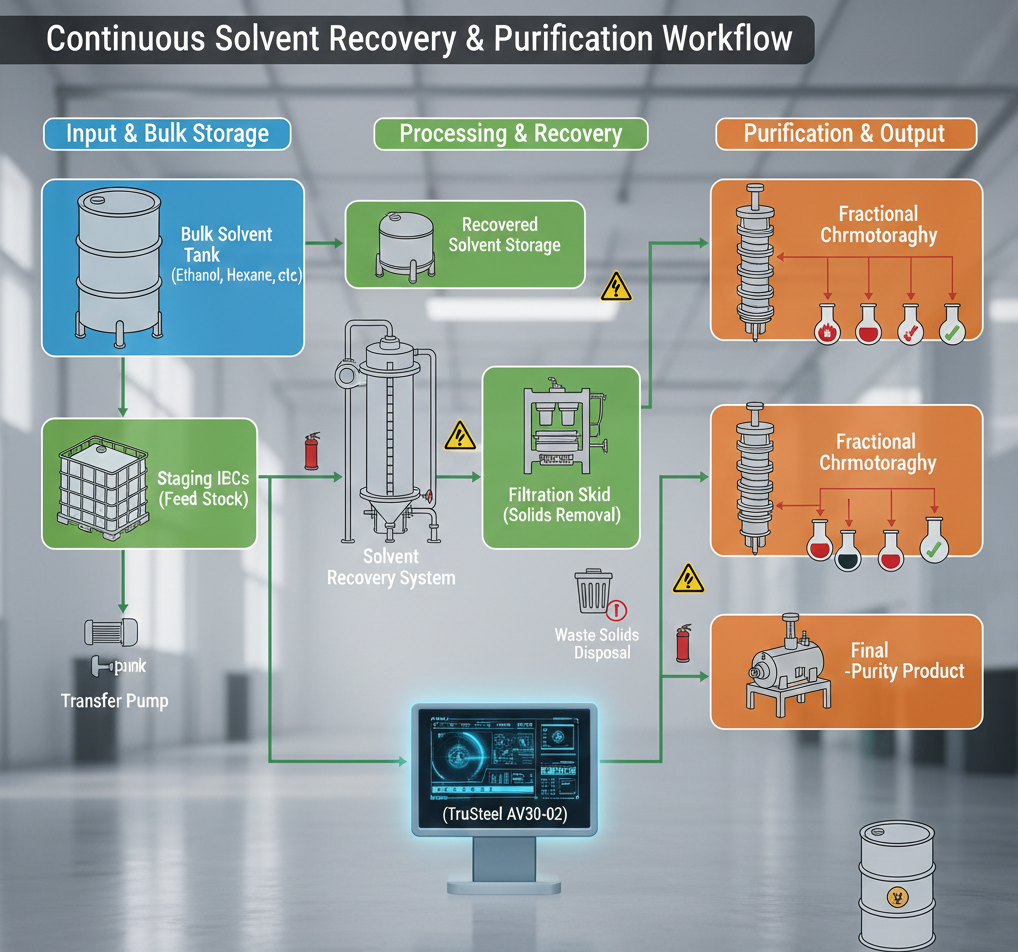
Mitigating Risk through Automated Precision
The high stakes of industrial manufacturing require a move away from the “trust-based” monitoring common in smaller labs. At production scale, automated control panels and real-time data monitoring are essential for mitigating the risk of expensive errors. Automated systems can track vacuum levels, temperature gradients, and flow rates with a level of precision that human operators cannot replicate consistently over 24-hour cycles.
This automation is particularly vital in fractional purification in chemical manufacturing when dealing with flammable materials or high-pressure environments. By utilizing localized control interfaces and emergency safety systems, facilities can detect deviations in the process before they escalate into significant losses. The ability to execute precise protocols, such as optimized orders of addition, ensures that the scale-up process is not only productive but inherently safe for personnel and assets alike.
Conclusion
Scaling a chemical process is an evolution from scientific discovery to industrial engineering. Mastering fractional purification in chemical manufacturing requires a balanced understanding of chemical kinetics and the complex logistics of bulk production. By recognizing that the primary bottlenecks at scale are operational rather than chemical, industry leaders can invest in the robust infrastructure and safety systems necessary for success. The true measure of a successful scale-up is the ability to manage the increased consequences of production with a data-driven, automated approach that ensures reliability and survivability at every stage.





