From Extraction to Engineering: The New Standard in Cannabinoid Science
The evolution of the cannabis industry from agricultural extraction to sophisticated pharmaceutical manufacturing has necessitated a shift toward advanced organic synthesis. At the heart of this transition is the concept of chirality. In medicinal chemistry, the spatial arrangement of atoms—the “handedness” of a molecule—can dictate whether a compound is a therapeutic breakthrough or a biological inert. Enantioselective hydrogenation has emerged as a critical methodology for chemists seeking to produce enantiomerically pure cannabinoids, ensuring that only the desired isomer is synthesized to interact with the human endocannabinoid system.
The Mechanics of Chiral Reduction and Selective Methodologies
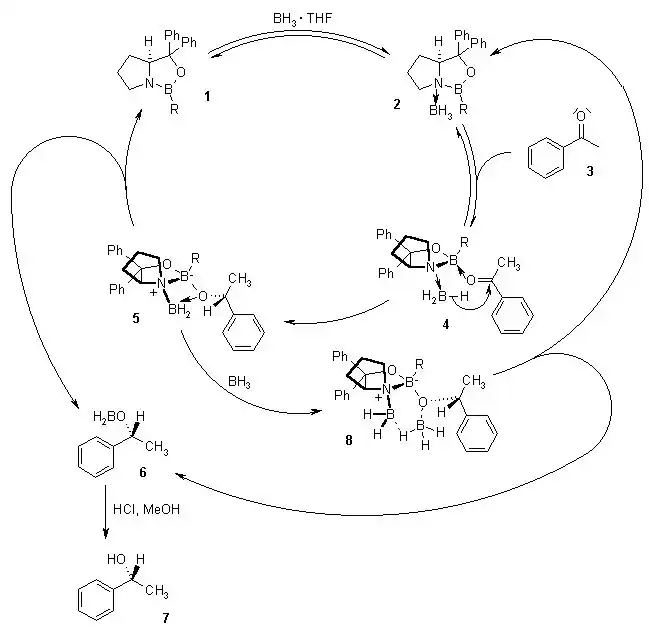
To achieve high enantiomeric excess (ee), laboratories utilize specific chemical pathways that favor the formation of one enantiomer over another. One of the most reliable methods is the Corey-Bakshi-Shibata (CBS) reduction. This process employs a chiral oxazaborolidine catalyst to reduce ketones. In the context of cannabinoid derivatives, the CBS reduction provides a predictable stereochemical outcome, which is essential for scaling up synthetic minor cannabinoids.
Another specialized approach is the Luche reduction, which utilizes cerium(III) chloride in conjunction with sodium borohydride. While traditionally known for the selective reduction of α,β-unsaturated ketones to allylic alcohols, its role in maintaining the integrity of the molecule’s backbone while altering specific functional groups is invaluable. For more direct stereochemical control, L-Selectride acts as a bulky, sterically hindered reducing agent, often used when the chemist must navigate complex molecular architectures where standard reagents would lack the necessary precision.
Transition Metal Catalysis and Ligand Engineering
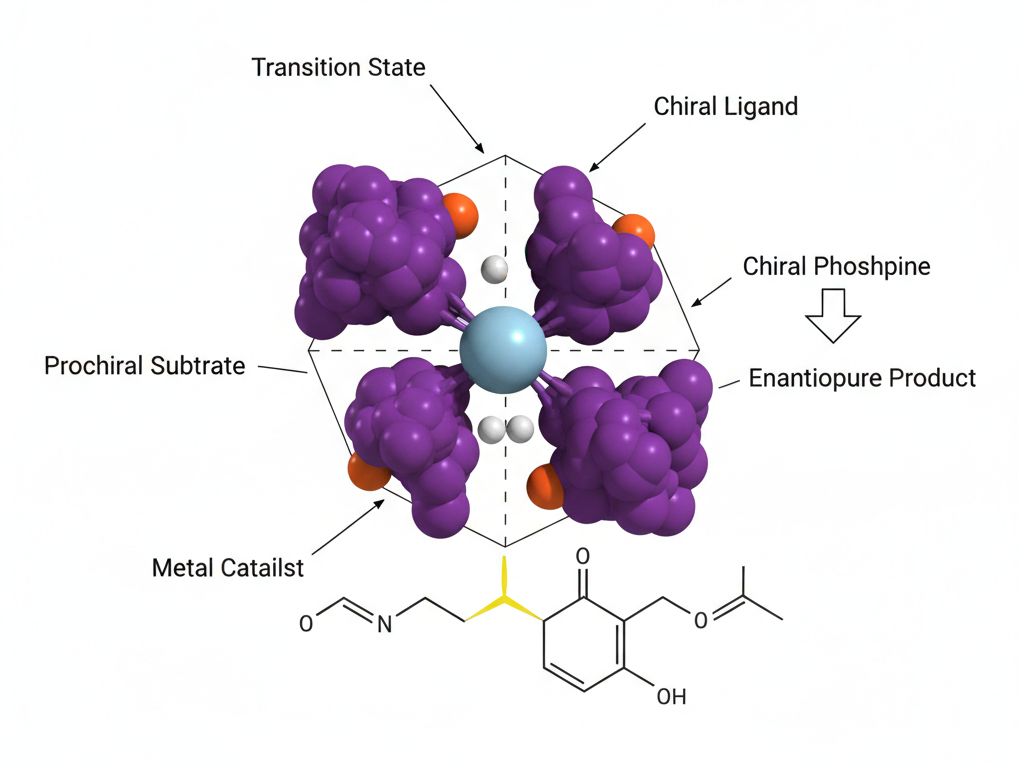
The most versatile tool for large-scale enantioselective hydrogenation is transition metal catalysis. By pairing metals such as rhodium, ruthenium, or palladium with sophisticated chiral ligands, researchers can hydrogenate prochiral olefins and ketones with remarkable efficiency. The ligand acts as a “chiral template,” guiding the hydrogen atoms to a specific face of the molecule.
While transition metal catalysis can be performed without chiral ligands, the result is typically a racemic mixture—an equal blend of left- and right-handed molecules. In the pharmaceutical-grade cannabinoid space, racemic mixtures are increasingly viewed as sub-optimal. The industry is moving toward “ligand-directed” synthesis because it allows for the creation of high-purity isolates that meet the rigorous standards of modern clinical research.
Pharmacological Implications of Enantiomeric Purity
The drive toward enantioselective hydrogenation is not merely a technical exercise; it is rooted in pharmacology. Most biological receptors, including CB1 and CB2, are themselves chiral. Consequently, the (R)-enantiomer of a cannabinoid may fit perfectly into a receptor pocket, while the (S)-enantiomer may be inactive or, in some cases, cause adverse effects.
By producing enantiomerically pure cannabinoids, researchers can enhance the efficacy of a compound while lowering the required dose. This precision reduces the metabolic load on the patient and minimizes off-target interactions. As we move toward targeted therapeutics for conditions like chronic pain, neuroinflammation, and epilepsy, the ability to control the “handedness” of the molecule becomes a primary safety requirement.
Future Horizons in Cannabinoid Therapeutics
The application of these high-level synthetic techniques marks the beginning of a new era in cannabinoid science. We are no longer limited to the ratios provided by the plant. Through enantioselective hydrogenation, the industry can explore the therapeutic potential of rare, minor cannabinoids and novel analogs that do not exist in nature.
This level of control over molecular geometry allows for the development of “designer” cannabinoids with specific pharmacokinetic profiles—adjusting how they are absorbed, distributed, and metabolized. As the regulatory landscape matures, the laboratories that master these enantioselective processes will be the ones leading the charge in pharmaceutical innovation and patented therapeutic discovery.