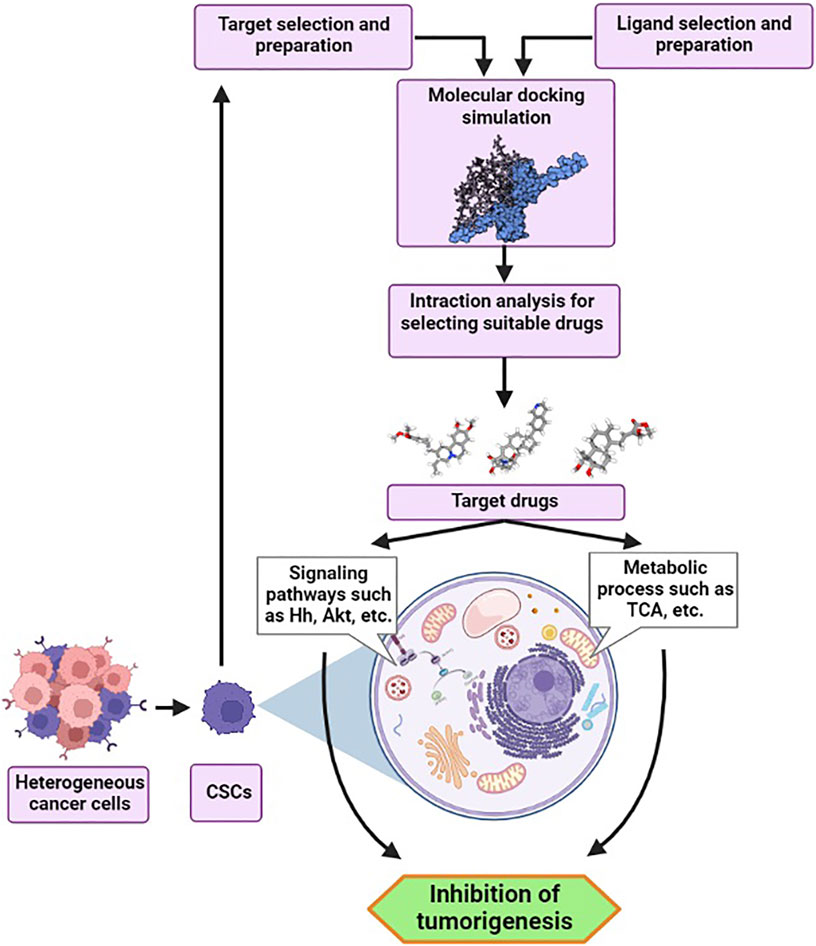
Introduction
Understanding how the human body transforms chemical compounds is essential in medicinal chemistry and pharmaceutical research. Any molecule introduced into a biological system is subject to metabolic processes that can alter its structure, activity, and safety profile. These transformations influence not only efficacy but also toxicity, exposure, and dosing frequency. As drug discovery increasingly relies on rational design, anticipating metabolic behavior early in development has become a scientific necessity rather than a late stage concern.
The Role of Metabolism in Structure Activity Relationships
Drug metabolism is primarily driven by enzyme systems that recognize and chemically modify foreign compounds. Among these systems, cytochrome P450 enzymes play a dominant role by oxidizing susceptible regions of a molecule. These reactions can deactivate a compound, generate reactive intermediates, or create active metabolites. From a structure activity relationship perspective, metabolism can obscure the true relationship between chemical structure and biological response. A compound may appear weak or unstable not because it lacks intrinsic potency, but because it is rapidly transformed in the body. Recognizing this distinction is critical when interpreting SAR data.
Predicting Metabolic Hotspots Using In Silico Tools
Computational modeling offers a powerful way to anticipate metabolic liabilities before extensive laboratory work begins. In silico approaches can identify chemically labile regions that are prone to oxidation or other transformations. By simulating how enzymes may interact with a compound, researchers gain early insight into metabolic stability and potential degradation pathways. These predictions allow scientists to prioritize analogs that balance target binding with favorable metabolic profiles, ultimately reducing wasted synthesis and testing cycles.
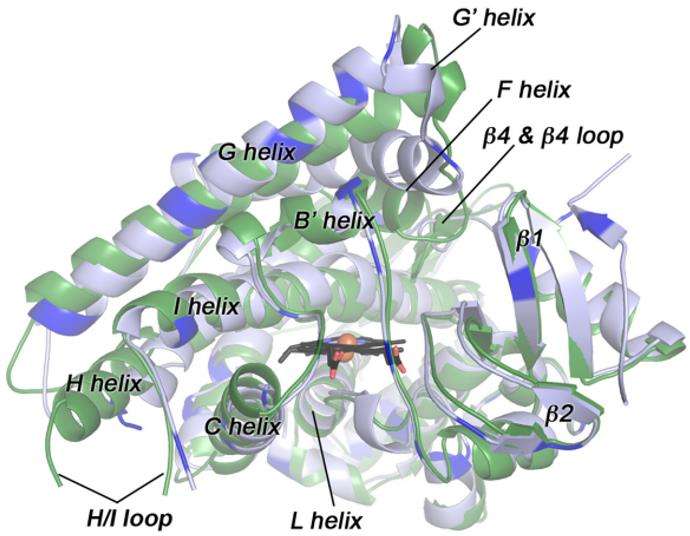
Stabilizing Key Binding Regions Through Rational Design
Certain therapeutics such as the tyrosine kinase inhibitor Gefitinib illustrate the importance of metabolic considerations in design. This class of molecules often contains functional groups that are vulnerable to enzymatic oxidation. If such a site is also essential for protein ligand interactions, uncontrolled metabolism can compromise therapeutic performance. Medicinal chemists can strategically modify these regions by introducing steric hindrance or electronic effects that reduce enzyme access while preserving binding affinity. This approach transforms metabolism from a limitation into a design parameter that can be actively optimized.
Conclusion
Metabolite awareness is a defining feature of modern drug discovery. By integrating metabolic predictions into SAR analysis, researchers can design compounds that are not only potent but also stable and durable in vivo. Computational tools provide a valuable preview of how molecules may behave biologically, enabling smarter design choices earlier in development. With thoughtful optimization, today’s SAR insights could indeed lead to tomorrow’s breakthrough therapies.