The Role of Catalytic Hydrogenation in Industrial Organic Chemistry
The transformation of unsaturated hydrocarbons into saturated frameworks is a fundamental requirement in modern synthetic chemistry. Catalytic hydrogenation serves as the primary mechanism for this conversion, specifically focusing on the reduction of olefins into alkanes through the addition of diatomic hydrogen. This process is not merely a laboratory curiosity but a foundational industrial operation that allows for the precise modification of molecular properties. By utilizing specialized metallic catalysts, chemists can achieve high levels of selectivity and efficiency, making this reaction a cornerstone of pharmaceutical development, petrochemical refining, and sustainable chemical manufacturing.
Fundamental Mechanics of Metal Mediated Hydrogen Addition
At its core, catalytic hydrogenation is a surface science phenomenon where a solid metal catalyst facilitates the reaction between a gas and a liquid or dissolved solid. Typically, transition metals such as palladium, platinum, or nickel are employed because of their unique ability to adsorb hydrogen molecules onto their surface. During this process, the hydrogen hydrogen bond is weakened or broken, creating active metal hydride species. Simultaneously, the olefin—characterized by its carbon carbon double bond—adsorbs onto the metal surface, positioning its pi system in close proximity to the activated hydrogen atoms.
The reaction proceeds through a sequential addition of hydrogen atoms across the double bond, effectively saturating the molecule and transforming it into an alkane. This reduction is highly valued because it is often clean, meaning it yields fewer byproducts compared to chemical reductions involving hydride reagents. In a laboratory setting, researchers often utilize specialized equipment like the Parr shaker or continuous flow reactors to maintain the necessary pressure and temperature, ensuring that the kinetics of the reaction are optimized for maximum throughput and purity.
Strategic Importance in Pharmaceutical and Fine Chemical Synthesis
In the pharmaceutical sector, the ability to selectively reduce double bonds is essential for the construction of Active Pharmaceutical Ingredients (APIs). Many natural products and synthetic intermediates contain unsaturated linkages that must be removed to achieve the correct molecular geometry or to enhance the metabolic stability of the drug. Catalytic hydrogenation provides a versatile tool for these transformations, allowing for the creation of saturated rings and chains that are more resistant to oxidative degradation within the biological environment.
Beyond simple reduction, advanced catalytic systems allow for chemoselectivity, where a double bond can be reduced in the presence of other sensitive functional groups such as nitriles, carbonyls, or halides. This precision is vital in multi step syntheses where maintaining the integrity of the broader molecular architecture is as important as the reduction itself. By carefully selecting the catalyst support and reaction solvent, chemists can tune the electronic environment of the metal site, driving innovation in the production of complex agrochemicals and high value fine chemicals.
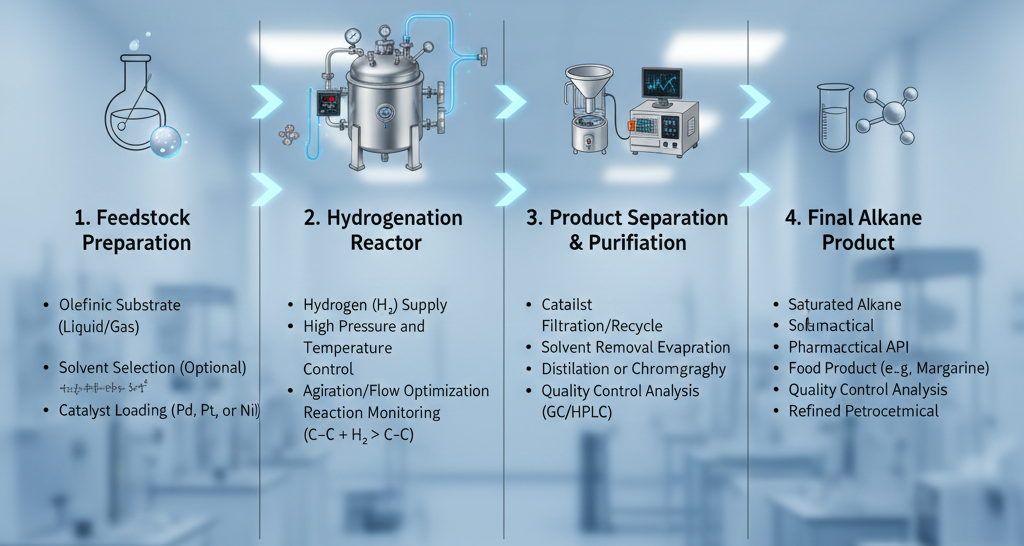
Industrial Scaling and the Push for Sustainable Chemistry
The industrial applications of hydrogenation are vast, ranging from the food industry to the refinement of fossil fuels. In the production of consumer goods, the partial hydrogenation of vegetable oils is used to modify the melting point and oxidative stability of fats, a process essential for the creation of various food products. In the petrochemical industry, hydrogenation is utilized in hydrocracking and hydrotreating processes to remove impurities like sulfur and nitrogen, as well as to saturate aromatics, resulting in cleaner burning fuels and more stable lubricants.
From a sustainability perspective, catalytic hydrogenation is often cited as a benchmark for green chemistry. Because the primary reagent is hydrogen gas, the atom economy of the reaction is exceptionally high, with nearly every atom of the starting materials ending up in the final product. Furthermore, as the industry moves toward green hydrogen produced via electrolysis powered by renewable energy, the carbon footprint of these industrial reductions continues to decrease. This transition allows chemical manufacturers to meet stringent environmental regulations while maintaining the high efficiency standards required for global production.
Conclusion
Catalytic hydrogenation of olefins represents a perfect synergy between theoretical chemical principles and large scale practical application. Its ability to simplify complex molecular structures while maintaining high yields and environmental compatibility makes it an indispensable asset in the chemist’s repertoire. As research continues into new catalyst geometries and sustainable hydrogen sources, the role of this reaction in driving industrial innovation and improving quality of life will only expand. For the modern researcher or industry professional, mastering the nuances of this catalytic process is essential for navigating the future of organic synthesis and chemical manufacturing.





