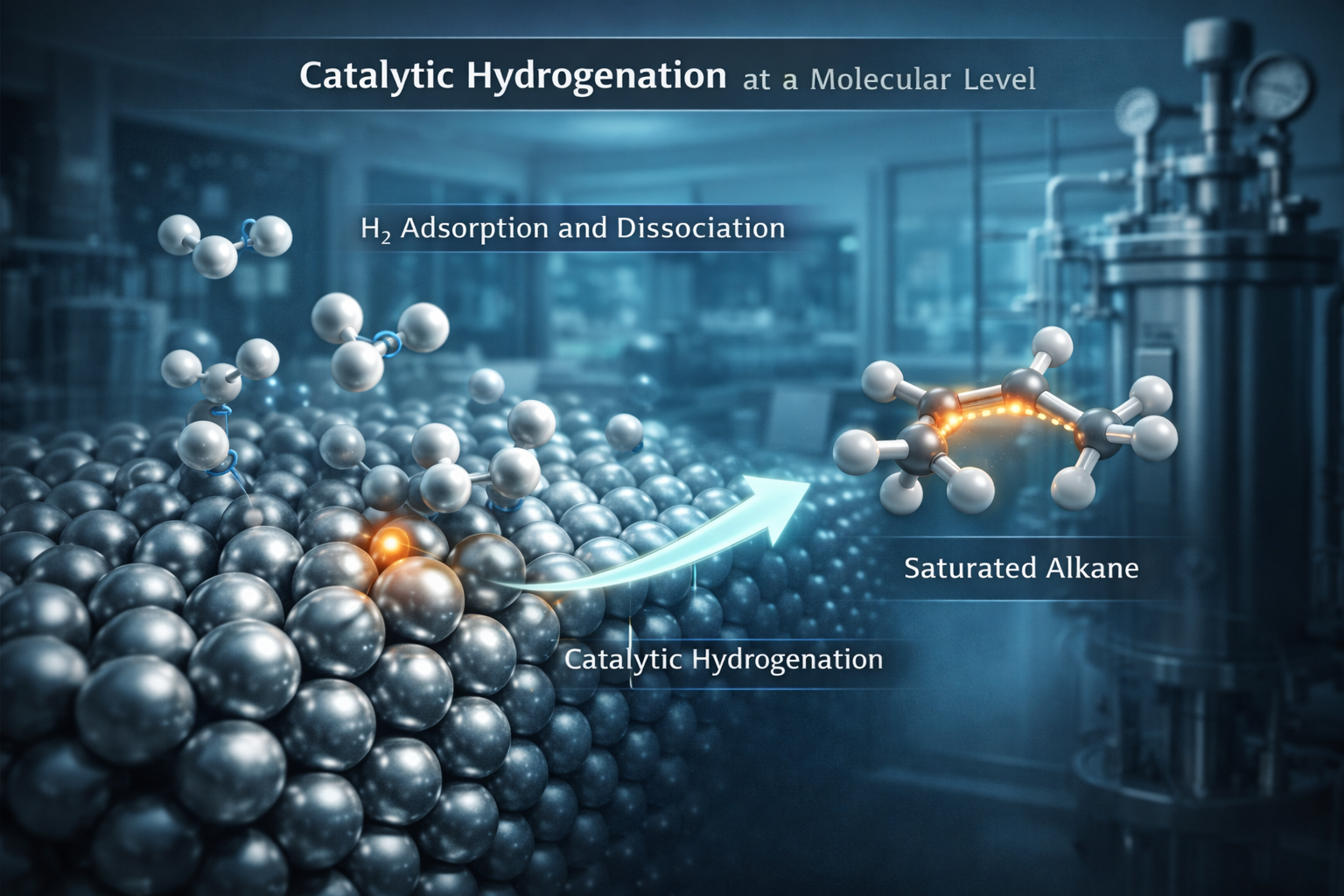
The transformation of unsaturated hydrocarbons into saturated frameworks remains a fundamental operation in synthetic organic chemistry. Catalytic hydrogenation serves as the primary mechanism for this conversion, facilitating the addition of diatomic hydrogen across carbon-carbon double bonds, or olefins, to produce alkanes. This chemical reduction is not merely a laboratory procedure but a cornerstone of modern industrial manufacturing. By utilizing specialized metallic catalysts, researchers and engineers can achieve high levels of precision, creating molecules with the specific stability and structural properties required for advanced applications. As global industries pivot toward more efficient and sustainable production methods, understanding the nuances of this reaction is essential for driving innovation in pharmaceutical and material sciences.
Mechanism and Metallic Catalysts in Surface Science
At the molecular level, catalytic hydrogenation is a heterogeneous process occurring at the interface of a solid metal surface and a fluid phase. The efficiency of the reaction is dictated by the choice of catalyst, typically transition metals such as palladium, platinum, or nickel. These metals possess a unique electronic structure that allows for the adsorption and activation of hydrogen molecules. During the process, the hydrogen-hydrogen bond is weakened as the atoms bind to the metal lattice, creating active metal-hydride species. Simultaneously, the olefin adsorbs onto the surface, positioning the pi-bond in close proximity to the activated hydrogen.
The reaction follows a sequential addition mechanism where hydrogen atoms are transferred to the carbon atoms of the double bond. This metal-mediated pathway significantly lowers the activation energy required for the reaction, allowing it to proceed under relatively mild conditions. In an industrial context, the ability to tune reaction parameters such as temperature, pressure, and catalyst support is vital for achieving high throughput and selectivity. Precise control over these factors ensures that the desired alkane is produced with minimal byproducts, a critical requirement for high-purity chemical manufacturing.

Strategic Role in Pharmaceutical and Agrochemical Development
The versatility of catalytic hydrogenation is most evident in the synthesis of complex bioactive molecules. In the pharmaceutical sector, this reaction is indispensable for the construction of Active Pharmaceutical Ingredients (APIs). Many drug candidates contain specific unsaturated linkages that must be reduced to achieve the correct molecular geometry or metabolic stability. By employing selective hydrogenation, chemists can target specific double bonds while leaving other sensitive functional groups intact, a level of precision that is fundamental to modern drug design.
Beyond pharmaceuticals, the agrochemical industry relies on this reaction to produce stable and effective pesticides and herbicides. The transition from olefins to alkanes often enhances the chemical stability of these compounds, ensuring they remain active for the intended duration when applied in the field. The reliability and scalability of catalytic hydrogenation make it a preferred method for creating a wide array of fine chemicals, where consistency in molecular structure is directly linked to the performance and safety of the final product.
Sustainability and Green Chemistry in Modern Industry
As the chemical industry faces increasing pressure to reduce its environmental footprint, catalytic hydrogenation stands out as a benchmark for sustainable chemistry. Unlike many chemical reduction methods that require stoichiometric amounts of hazardous reagents and generate significant waste, hydrogenation utilizes diatomic hydrogen gas. When hydrogen is sourced through sustainable methods, such as water electrolysis powered by renewable energy, the overall environmental impact of the synthesis is drastically reduced.
The high atom economy of the reaction, where nearly every atom of the starting materials ends up in the final product, aligns perfectly with the principles of green chemistry. Furthermore, the ability to recover and reuse expensive transition metal catalysts like platinum or palladium adds to the economic and environmental viability of the process. In industries ranging from food production—specifically the hydrogenation of vegetable oils to produce stable fats—to petrochemical refining and polymer synthesis, this reaction continues to facilitate cleaner, more efficient manufacturing pathways.
Conclusion
Catalytic hydrogenation of olefins represents a sophisticated marriage of fundamental surface science and practical industrial utility. Its role in simplifying complex syntheses while maintaining high levels of safety and environmental compatibility makes it a permanent fixture in the chemist’s repertoire. As we look toward the future, the continued refinement of catalyst technologies and the integration of sustainable hydrogen sources will further enhance the value of this reaction. For researchers and industry experts alike, mastering the application of catalytic hydrogenation is essential for developing the next generation of materials and therapeutics that will define the quality of life in the years to come.